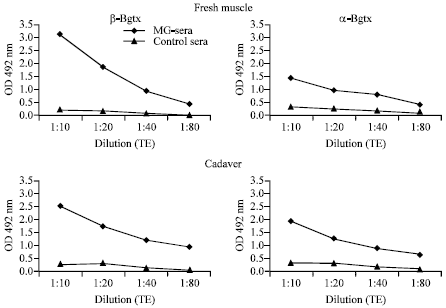
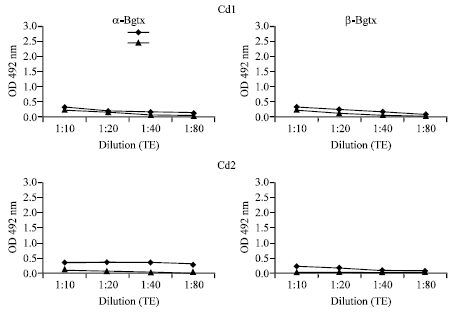
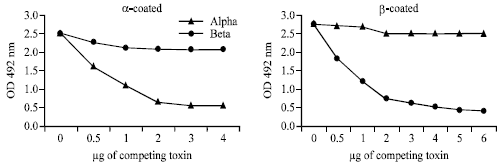
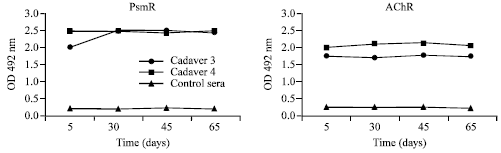
Research Article
Antibodies Against Postsynaptic Acetylcholine Receptor and Presynaptic Membrane Receptor in Myasthenia Gravis
Department of Laboratory Medicine,New Delhi, 110029, India
Madhuri Behari
Neurosciences Center, Department of Neurology, All India Institute of Medical Sciences, New Delhi, 110029, India
M. Irshad
Department of Laboratory Medicine,New Delhi, 110029, India
Vinay Goyal
Neurosciences Center, Department of Neurology, All India Institute of Medical Sciences, New Delhi, 110029, India
B.L. Jailkhani
Department of Laboratory Medicine,New Delhi, 110029, India