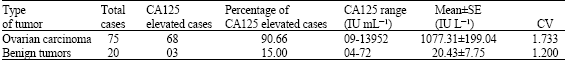Research Article
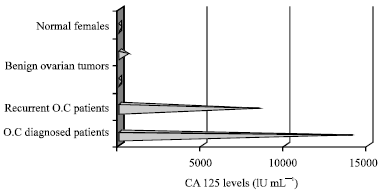
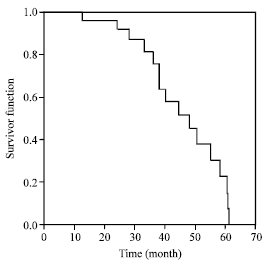
Specificity of Serum Tumor Markers (CA125, CEA, AFP, Beta HCG) in Ovarian Malignancies
Department of Biochemistry, Indo-American Cancer Institute and Research Centre, RoadNo.l4, Hyderabad500 034, AP. India
Kaiser Jamil
Department of Biochemistry, Indo-American Cancer Institute and Research Centre, RoadNo.l4, Hyderabad500 034, AP. India