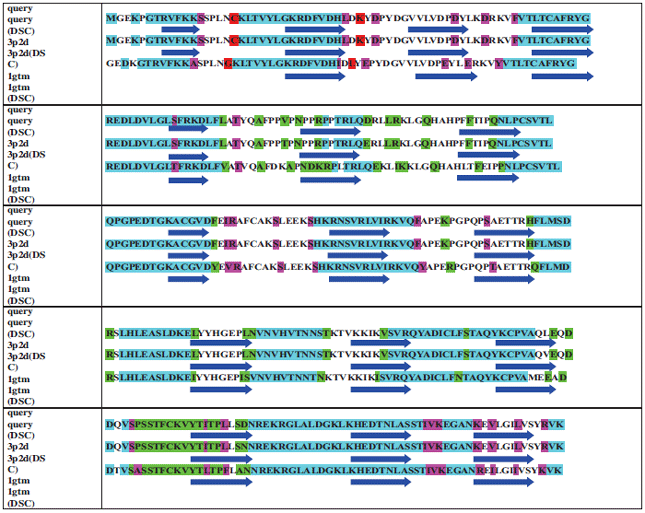
Research Article
Study on Role of Homology in Prediction of Protein Structure and Challenges of Homology
Department of Plant Science, College of Agriculture and Veterinary Sciences, Ambo University, Post Box: 19, Ambo, Ethiopia, East Africa
LiveDNA: 91.10338