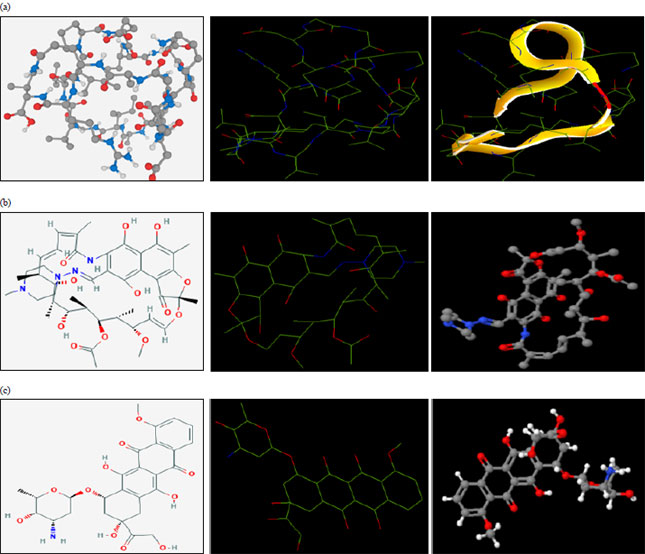
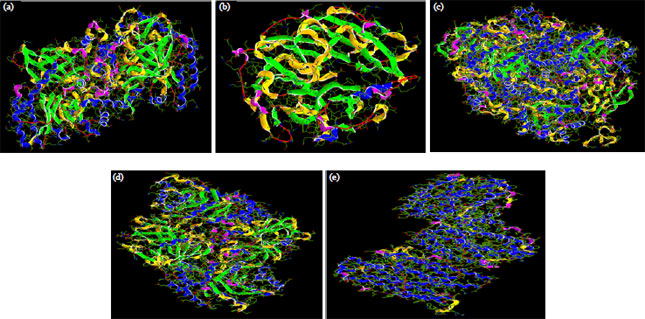
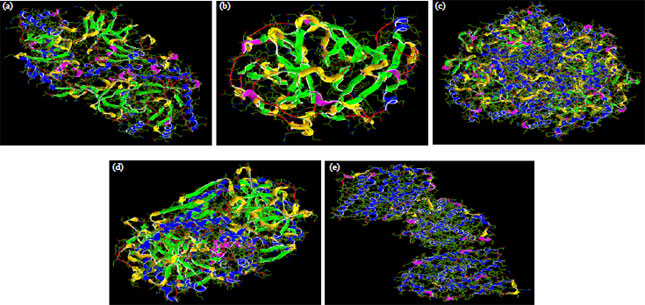
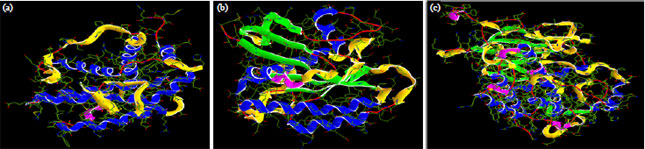
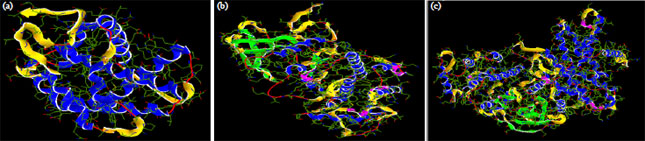
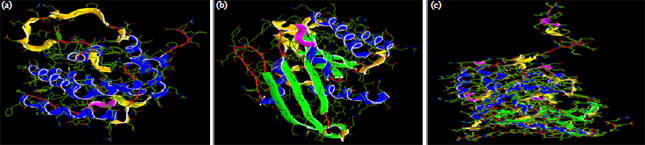
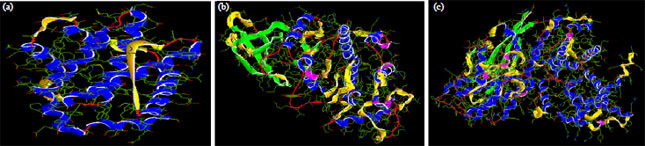
Research Article
In silico Modelling and Molecular Docking Insight of Bacterial Peptide for Anti-tubercular and Anticancer Drug Designing
Research Department of Plant Biology and Biotechnology, Loyola College, 34 Chennai, Tamil Nadu, India
LiveDNA: 91.8089
Chirom Aarti
Research Department of Plant Biology and Biotechnology, Loyola College, 34 Chennai, Tamil Nadu, India
Paul Agastian
Research Department of Plant Biology and Biotechnology, Loyola College, 34 Chennai, Tamil Nadu, India