Research Article
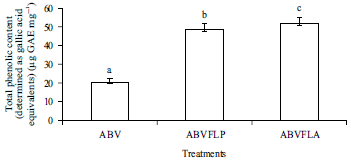
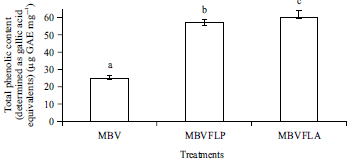
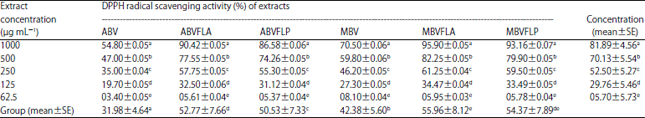
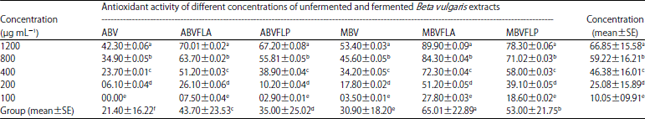
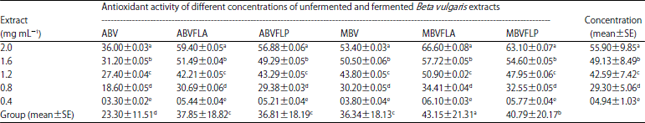
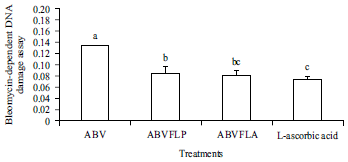
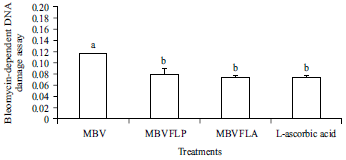
Enhancement of Antioxidant Activity, Phenolic Contents and Protective Effects of Beta vulgaris Root Extract Against DNA Damage by Fermentation using Lactic Acid Bacteria
Department of Microbiology and Immunology, Faculty of Pharmacy, Zagazig University, Zagazig, Egypt
LiveDNA: 20.28622
Mahmoud A. Al-Saman
Department of Industrial Biotechnology, Genetic Engineering and Biotechnology Research Institute, University of Sadat City, Sadat City, Egypt
LiveDNA: 20.21831
Asmaa Abdella
Department of Industrial Biotechnology, Genetic Engineering and Biotechnology Research Institute, University of Sadat City, Sadat City, Egypt
LiveDNA: 20.21679
Hoda Mahrous
Department of Industrial Biotechnology, Genetic Engineering and Biotechnology Research Institute, University of Sadat City, Sadat City, Egypt
LiveDNA: 20.3942