Research Article
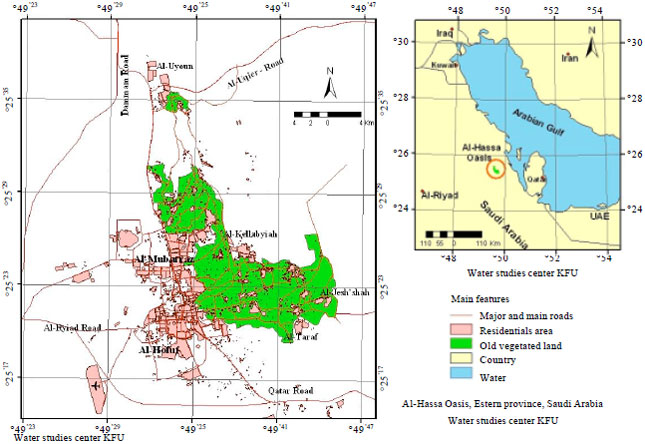
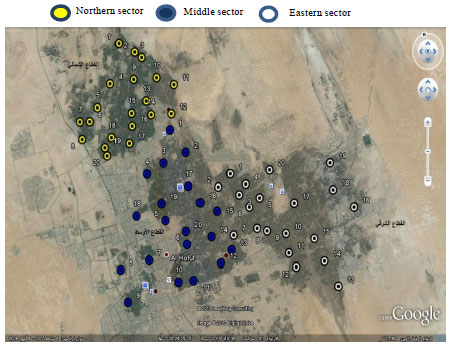
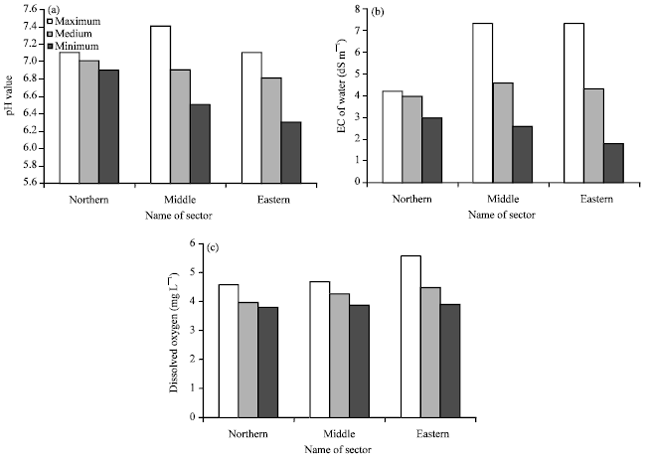
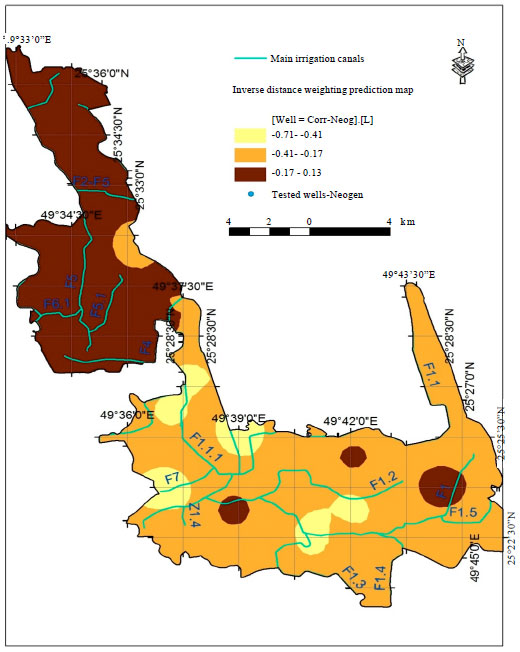
Spatial Pattern of Groundwater Quality and its Impact on Pipes and Wells Equipment Corrosion in Al-Hassa Oasis Kingdom of Saudi Arabia
Department of Environment and Natural Agricultural Resources, College of Agricultural and Food Sciences, King Faisal University, Al-Hassa, Kingdom of Saudi Arabia
A.A. Al- Naeem
Department of Environment and Natural Agricultural Resources, College of Agricultural and Food Sciences, King Faisal University, Al-Hassa, Kingdom of Saudi Arabia