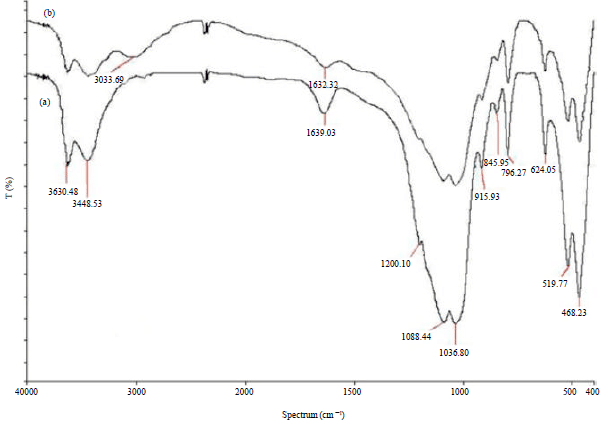
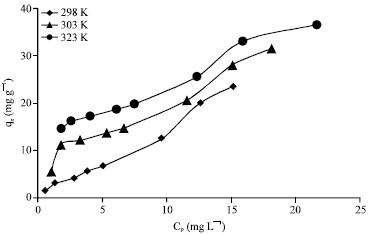
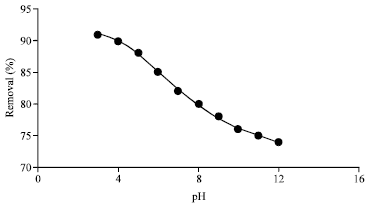
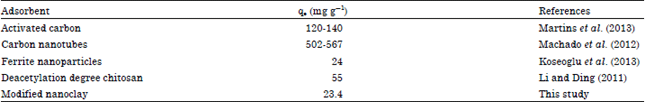Research Article
Adsorption Behavior of Reactive Blue 29 Dye on Modified Nanoclay
Department of Environmental Health Engineering, School of Health, Yasuj University of Medical Sciences, Yasuj, Iran
Mohammad Rafiee
Department of Environmental Health Engineering, Alborz University of Medical Sciences, Alborz, Iran
Mahsa Jahangiri Rad
Department of Environmental Health Engineering, Islamic Azad University, Tehran Medical Branch, Tehran, Iran