Research Article
Thermodynamic and Polymeric Inhibitors Effects on Methane Gas Hydrate Formation
Department of Mechanical Engineering, Science and Research Branch, Islamic Azad University, Arak, Iran
Gas hydrates are ice like crystalline compounds consisting of low molecular diameter gases inside cavities formed by water molecules which can form at certain pressure and relatively low temperature conditions. They are a subset of compounds known as clathrates or inclusion compounds (Carroll, 2002). A hydrocarbon hydrate generally is composed of crystals formed by water host molecules surrounding the hydrocarbon guest molecules. The water network is stabilized by weak Van der Waals forces between the host and the internal guest molecules (Gaudette and Servio, 2007). The smaller or lower-boiling hydrocarbon molecules, particularly C1 to C4 hydrocarbons and their mixtures, are more problematic because it is believed that their hydrate or clathrate crystals are easier to form.
The oil and gas industry are continuously looking for hydrate inhibitors that will allow them to venture out into deeper waters and operate their processes without the risk of hydrates interfering with daily operations. There are two classes of hydrate inhibitors; thermodynamic and kinetic inhibitors. For the thermodynamic approach, there are a number of attempted methods, including water removal, increasing temperature and/or decreasing pressure (favorite conditions for hydrate formation), addition of antifreeze to the fluid and/or a combination of these (Zhang et al., 2004; Mokhatab et al., 2007; Najibi et al., 2009). The kinetic approach generally attempts to prevent the smaller hydrocarbon hydrate crystals from agglomerating into larger ones and to inhibit and/or retard initial hydrocarbon hydrate crystal nucleation and/or crystal growth. Thermodynamic and kinetic hydrate control methods may be used in conjunction.
A common method to prevent hydrate formation is to use large quantities of thermodynamic inhibitors such as methanol and glycols. The addition of a sufficient amount of these compounds moves the conditions required for hydrate formation to lower temperatures and/or higher pressures (Gokturk and Var, 2011). However, as temperature and pressure conditions in the field become more severe (e.g., as exploration and production moves to deeper waters) the concentration of inhibitor required to prevent hydrate formation increases dramatically, often in excess of 60 wt.% but are costly in offshore developments and onshore processing facilities (Koh et al., 2002; Lovell and Pakulski, 2003; Lee and Englezos, 2006; Zeng et al., 2008). These are water-soluble chemicals that reduce the water activity, hence shifting the hydrate phase boundary to higher pressure and/or lower temperature conditions (Koh et al., 2002). In general, produced water contains various quantities of salts, reducing the tendency for gas hydrate formation. In cases where the inhibition effect of the produced saline water is not sufficient to prevent hydrate formation, thermodynamic inhibitors could be injected into the pipelines, resulting in a system containing both salts and inhibitor (Najibi et al., 2009).
Kinetic inhibition methods are based on the injection of polymer-based chemicals at low dosages in the water phase (Lee and Englezos, 2006). Kinetic Hydrate Inhibitors (KHIs) delay hydrate nucleation and usually also crystal growth. These inhibitors bond to the hydrate surface and prevent plug formation for a period longer than the free water residence time in a pipeline. Liquid hydrocarbons may or may not be present for this prevention method to be effective. Kinetic inhibitors are limited at long times, low temperatures and high pressures because with sufficient time, the crystal growth is significant enough to cause line plugs (Huo et al., 2001; Lovell and Pakulski, 2003; Kelland et al., 2006; Sloan and Koh, 2008). Additives such as polymers with lactam rings have also been employed to control clathrate hydrates in fluid systems. These kinetic inhibitors are commonly labeled Low Dosage Hydrate Inhibitors (LDHI) in the art. The common KHIs are: polyvinylpyrrolidone (PVP) and polyvinylcaprolactam (PVCap) (Suliman et al., 2006; Al-Adel et al., 2008; Anklam and Firoozabadi, 2005). These kinetic inhibitors are effective at significantly lower concentrations (0.1-1.0 wt.%) respect to the thermodynamic inhibitors concentration (40-60 wt.%) (Sloan and Koh, 2008). Figure 1 illustrates the chemical structure of PVP.
The hydrate nucleation in gas hydrate crystallization is an important characteristic of the kinetics of the process (Kashchiev and Firoozabadi, 2003). Hydrate nucleation also called the induction period is process where small hydrate crystals nuclei grow and disperse until they reach a critical size or radius for continuous growth (Bishnoi and Natarajan, 1996). The nucleation time or induction time is measured during each hydrate formation experiment. It is defined as the time where the stirrer was turned-on in the reactor until hydrates are observed which indicates that the growth period has started (Al-Adel, 2007). The reactor liquid temperature is useful to determine the nucleation time since a spike in the temperature is usually observed during the initial stage of growth (Fig. 2) (Al-Adel et al., 2008). Nucleation times are compared between experiments especially when inhibitors are used to check their effect on induction period. Hydrate growth refers to the growth of stable hydrate nuclei as solid crystals after they reach the critical size (Bergeron and Servio, 2008).
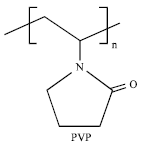 | |
| Fig. 1: | The structure of poly[N-vinylpyrrolidone], PVP[13] |
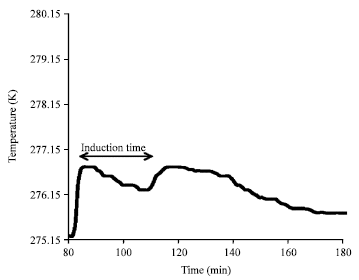 | |
| Fig. 2: | Change in the temperature during methane hydrate formation at T ≈ 276.6 K and p = 56 bar |
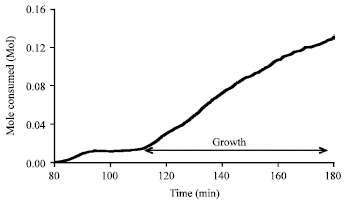 | |
| Fig. 3: | Methane consumed during hydrate formation at an initial temperature of 276.6 K and pressure of 60 bar for deionized water |
The growth periods measured for deionized water were considered as a reference point (Fig. 3). Then the growth periods measured for the other solutions were compared to the reference in order to determine their effect on growth.
The purpose of this study was to investigate the effects of two kinds of inhibitors (thermodynamics and kinetic inhibitors) on Methane hydrate formation process (on the percentage of inhibition and induction time). For thermodynamic inhibitor, Calcium Chloride, Three Ethylene Glycol and Methanol with 5 weight percent and PVP for kinetic inhibitor with 0.1, 0.2 and 0.5 weight percent were tested. Also the impact of Polyethylene Oxide (PEO) on the performance of kinetic inhibitor (PVP) was demonstrated.
Materials: Fluid samples used in the experiments were containing deionized water, methane (99.5%, Technical Gas Services Co.), methanol (99 mol%, Acros Organics), triethylene glycol (TEG) (99 mol%, Acros Organics), calcium chloride (CaCl2) (Merck) and PVP (dry powder with Mw = 8000, Acros Organics).
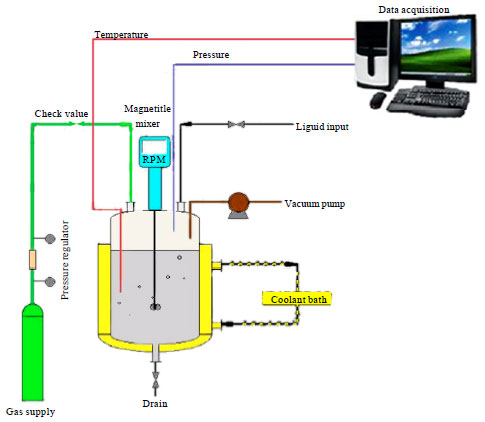 | |
| Fig. 4: | A schematic diagram of the hydrate formation setup |
Apparatus: The schematic diagram of the experimental apparatus used is shown in Fig. 4. It consisted of a high-pressure stainless steel crystallizer with a total volume of 1180 cm3 is used to produce the gas hydrate. The reactor was equipped with a magnetic stirrer with speed 500 rpm for agitation of the liquid phase.
One platinum resistance thermometer (Pt100) with an accuracy of ±0.1 K and pressure transducer (Huba control-5436 worenlos model) with an accuracy of ±0.1 bar inserted into the vessel were used to measure temperature and pressure, respectively. The pressure and temperature of the system are continuously recorded on the computer during the experiments. The reactor had valves for injecting and venting the gas as well as vacuum pump. The temperature in the cell was controlled by circulating coolant (water-ethylene glycol solution), in a jacket surrounding the cell.
Procedure: The crystallizer was cleaned prior to the injection of a new solution. The solution was either deionized water only which was set as a reference point or included a polymer, salt and/or alcohol. The 500 cm3 of various solutions were injected into the crystallizer. The vacuum pump was turned on and allowed the air inside the reactor to come out. The air inside the reactor was flushed out by repeating pressurizations with methane gas up to 10bar and depressurizations to atmospheric pressure two times. Subsequently, crystallizer is filled with the methane gas until the desired pressure is obtained. As soon as the temperature and pressure stabilized, the experiment commenced and data acquisition system were started. The first indicated time was zero. The bath temperature was reduced till 273.6 K and the solution was kept for 90 min without any mixing. After that stirrer starts rotating with 500 rpm. The recorded pressure drop in the crystallizer during the experiment was used to calculate the number moles of gas entering the crystallizer. This is called the consumed moles and can directly be measured by the consumed gas calculation for hydrates formation during an experiment, by using real gas law; PV = nZRT that P, V and T are the gas pressure, volume and temperature, respectively,. R is the gas constant and Z is the compressibility factor is calculated by Peng-Robinson equation of state (Al-Adel, 2007). In this study, the analysis and results included only the growth period. The mole consumption during the growth period was highlighted for the solutions that included the inhibitor and was compared with pure deionized water. The experiments were conducted in a methane-water system.
By carrying out experiment related to different kinds of inhibitors and deionized water it would be observed that the profile of hydrate growth is considerably influenced by kind and concentration of hydrate inhibitor. Table 1 shows carried experiments in this study as well as consumed inhibitor, percent of inhibition and induction time of different solutions. First experiment is related to solution without inhibitor (pure water) which is used as basis point for comparing with other inhibitors. In these experiments the effect of thermodynamic and polymeric inhibitors on the percentage of inhibition and induction time are studied.
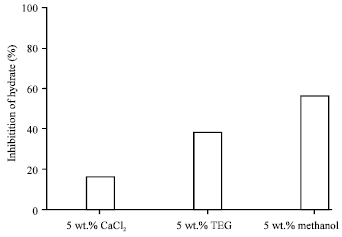
According to the thermodynamic inhibitors, induction time was the smallest value for 5 wt.% CaCl2 solution while it was the greatest value for methanol solution in the same as weight percent. These experiments show that methanol inhibitor delays induction time of methane hydrate more than other thermodynamic inhibitors. In addition, the percentage of inhibition of thermodynamic inhibitors after 30 min of hydrate growth is methanol>TEG>CaCl2.
In polymeric inhibitors, an increasing in the inhibitor concentration increases induction time. As shown in Table 1 induction time is around 106 min for 0.5 wt.% PVP solution.
Inclusion of polyethylene oxide (PEO) into a kinetic inhibitor solution was found to enhance the performance of the inhibitor. Polyethylene oxide is a commercially available high molecular weight polymer that is not a kinetic inhibitor by itself (Lee and Englezos, 2005). The enhanced inhibition effect of using this polymer on increasing time of methane hydrate formation for 0.1 and 0.5 wt.% solutions of PVP inhibitor can be seen in Table 1.
Induction time for 0.1 wt.% PVP solution is 7 min and for 0.5 wt.% PVP solution 106 min. Increasing 0.025 wt.% of PEO increases these times to 17 and 225 min correspondingly that indicates the increase of efficiency of PVP kinetic inhibitors in the present of PEO polymer.
Figure 5 shows methane mole consumed rate in 30 min after the hydrate formation in the present of 5 wt.% solution of methanol, TEG and CaCl2 salt at 277.6 K and 60bar.
| Table 1: | Experiment conditions and consumed rate after 30 min of hydrate growth in the presence of inhibitors |
 | |
 | |
| Fig. 5: | Methane mole consumed after 30 min of hydrate formation with thermodynamic inhibitors at an initial temperature of 276.6 K and pressure of 60 bar |
 | |
| Fig. 6: | Hydrate inhabitation percentage after 30 min of hydrate formation with thermodynamic inhibitors at an initial temperature of 276.6 K and pressure of 60 bar |
As it can be seen mole consumed of thermodynamic inhibitors of deionized water is smaller that indicates the effect of these solution in compare to deionized water. The amount of methane mole consumed by hydrate for methanol solution of 5 wt.% after 30 min of hydrate growth is 0.025 moles that its inhibition percent in proportion to deionized water would be about 56%. Comparing mole consumed by methane gas in the presence of thermodynamic inhibitors, the maximum percentage of inhibition is related to methanol solution of 5 wt.% (Fig. 6).
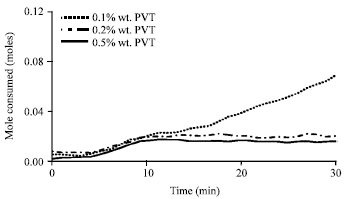
Figure 7 shows the methane mole consumed after 30 min of hydrate growth in different concentrations of PVP polymer inhibitor. By increasing PVP polymer concentration, mole consumed by methane hydrate has descending process and as a result the percent of inhibition increases. So that in a solution involving 0.5 wt.% PVP polymer we would have the maximum percent of hydrate inhibition which is 74%. In Fig. 8, methane hydrate inhibition percentage which increases with PVP polymer concentration is seen in the form of bar chart.
In order to examine PEO polymer effect on PVP polymer inhibitor, a solution including 0.025 wt.% PEO polymer+0.1 and 0.5 wt.% PVP solution are tested. As it can be seen in Fig. 9, adding PEO polymer shows the amount of hydrate inhibition in compare with when PVP polymer is used alone in two different concentrations and adding this polymer increases the percent of hydrate inhibition.
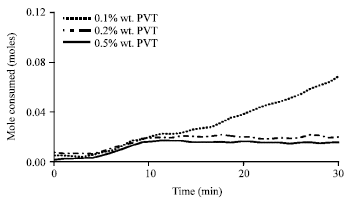 | |
| Fig. 7: | Methane mole consumed after 30 min of hydrate formation with polymeric inhibitor at an initial temperature of 276.6 K and pressure of 60 bar |
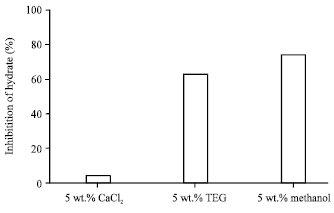 | |
| Fig. 8: | Hydrate inhabitation percentage after 30 min of hydrate formation with different concentration of polymeric inhibitor at an initial temperature of 276.6 K and pressure of 60 bar |
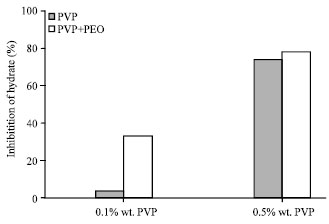 | |
| Fig. 9: | Hydrate inhabitation percentage after 30 min of hydrate formation with different concentration of polymeric inhibitor and PEO at an initial temperature of 276.6 K and pressure of 60 bar |
In Fig. 10, PEO effect on hydrate formation time can be seen, based on this figure, time of methane hydrate formation in the present of PVP inhibitor equals 188 min and with increasing PEO polymer this has increased to 335 min.
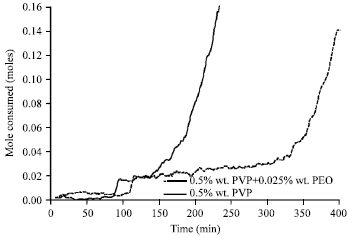 | |
| Fig. 10: | Methane mole consumed after 30 min of hydrate formation with polymeric inhibitor and PEO at an initial temperature of 276.6 K and pressure of 60 bar |
So PEO causes the increase of efficiency of kinetic and polymer inhibitors.
Experiments have been carried out to investigate the inhibition effects of thermodynamic and polymeric inhibitors on the Methane hydrate formation. For comparison, the experiments were performed at three types of thermodynamic and one type of polymeric (PVP) inhibitors along with a pure Methane-water system. In these experiments the effect of thermodynamic and polymeric inhibitors on the percentage of inhibition and induction time have been studied. Also Inclusion of polyethylene oxide (PEO) into a kinetic inhibitor solution as enhancing the performance of the kinetic inhibitor has been investigated.