Research Article
Microencapsulation of Colistin Sodium Methanesulfonate in Gum Arabic and Maltodextrin by Spray Drying
Department of Biochemistry, University of Benin, P.M.B. 1154, Benin City, Nigeria
Two antibiotics that are commonly aerosolized to manage Pseudomonas infections in Cystic Fibrosis (CF) patients are tobramycin and colistin (Ratjen et al., 2006). It is reported that inhaled tobramycin usually exceeds the minimal inhibitory concentration in the lungs and there is a low systemic absorption of inhaled tobramycin; hence systemic adverse effects rarely occur. Pseudomonas aeruginosa is naturally resistant to most antibiotics and thus its eradication does not usually occur with inhaled tobramycin (LoBue, 2005; Moskowitz et al., 2008; Sharifi-Yyazdi et al., 2007). Conversely, resistance to colistin, which is administered in two forms viz: colistin sulphate and Colistin Sodium Methanesulphonate (CMS), is rare and is thus the antibiotic of choice in the treatment of multidrug-resistant Pseudomonas aeruginosa strains (Bergen et al., 2006; Wootton et al., 2005).
Researchers are currently exploring aerosolization of antibiotics as a means to reduce dosage time and also to achieve high concentrations of these antibiotics at endobronchial sites of infection while limiting systemic bioavailability (Conway et al., 2003; Geller et al., 2002). It has been asserted that the most successful application of inhaled antibiotics has been for the management of Pseudomonas aeruginosa infections in CF patients. The commonest way to achieve this targeted delivery uses nebulizers to deliver the aerosolized drug (Pilcer et al., 2009). However, there are numerous disadvantages associated with the use of nebulizers for inhalation therapy, some of which include low patient compliance due to the requirement for repeated and prolonged treatment time, high cost, risk of microbial contamination and the need for bulky compressors or gas cylinders (Adi et al., 2008).
An alternative and more effective approach to the use of nebulizers in targeted inhalation therapy involves the administration of a Dry Powder Inhalation (DPI) product of the drug which is activated and driven by the patient’s inspiratory flow (Pilcer et al., 2008). Some of the technologies that have been employed to generate DPI microparticles by encapsulation include coacervation, solvent extraction, electrospray and spray drying (Xie and Wang, 2007).
Microencapsulation by spray drying is rapid, easy to scale up and less dependent on the solubility properties of the substance (Jain, 2000). Studies by Desai and Park (2005) asserted that spray drying is widely used by pharmaceutical industries to produce microparticles of heat-resistant, heat-sensitive, water-soluble or water-insoluble drugs in hydrophilic or hydrophobic polymers (Desai and Park, 2005). Spray drying is a single-step process that converts a liquid-a solution, emulsion or suspension-into a powder by atomization in a hot gas (Gharsallaoui et al., 2007). The sole aims of spray drying a compound are to generate microparticles of the substance with a range of particle size and also to remove most of the solvent so as to achieve a dry dosage form with long-term stability (Krishnaiah et al., 2009; Vehring, 2008). The gas that is commonly used is air but nitrogen can also be used (Patel et al., 2008).
The success of any encapsulation process is critically dependent on the choice of wall materials (Davidov-Pardo et al., 2008). Some of the polymers that have been used in spray drying processes include poly D-lactide, poly ε-caprolactone, poly (lactide co-glycolide) (Blanco et al., 2003; Blanco et al., 2005). However, organic solvents like dichlorometane is required for the formulation of the solution to be spray dried when these polymetric matrices are used (Bittner et al., 1999). This limitation is overcome by using carbohydrates which can form aqueous solutions. In addition, carbohydrates are good polymetric matrices for encapsulation because they are non-toxic, biocompatible, readily degraded after administration and have enormous potentials for controlled drug release (Sham et al., 2004; Surini et al., 2009).
In the food and pharmaceutical industries, gum arabic is used as a very effective emulsifier and stabilizing agent (Dror et al., 2006). In the flavor industry, gum arabic is used to encapsulate flavours thus protecting them from volatilization and oxidation (Krishnan et al., 2005). Another property of gum acacia that is employed by industries is its ability to adopt a compact spheroidal conformation due to its highly branched structure (Tombs and Harding, 1998). Another carbohydrate that can be employed in microencapsulation is maltodextrins which have numerous properties that allow them to be used for diverse purposes in both the food and pharmaceutical industries (Toure et al., 2007). Their ability to retain water and form gels explains their choice as efficient food stabilizers (Chronakis, 1998). They have also been used to stabilize spray-dried cellulases for long periods of time due to their ability to decrease water activity (Belghith et al., 2001). It has also reported that maltodextrins have been used as stabilizers in the microencapsulation of vitamins, minerals, colorants as well as fat and oils. However, the poor film-forming ability as well as hygroscopicity and turbidity of maltodextrins account for their inability to protect volatile compounds during spray drying (Raja et al., 1989).
There is a dearth of information on the microencapsulation of CMS by spray drying. Hence, the rationale of this study is to employ spray drying to generate a free-flowing Dry Powder Inhalation (DPI) formulation of CMS in the presence of a biodegradable polymer (gum arabic) wherein the antibiotic is protected and a slow continuous release of the drug might be ensured. Sodium chloride was also used as an excipient with the hope of generating biologically active microparticles that contain both the antibiotic and sodium chloride since the later has been proven to improve mucocilliary cleareance in cystic fibrosis patients (Elkins et al., 2006). The objectives of this study were to spray dry CMS in the presence of maltodextrin and sodium chloride; employ the disc diffusion assay to determine the residual anti-bactericidal activity of the spray-dried microparticles against a strain of Escherichia coli as well as to employ scanning electron microscopy to determine the surface morphology of the spray-dried particles.
Materials: Colistin sodium methanesulphonate (CMS) (CAS 8068 28-8; 11,500 Units mL-1) was purchased from Sigma-Aldrich® (Gillingham, UK). The indicator microorganism-Escherichia coli NCIMB 8879 was purchased from NCIMB Ltd (Aberdeen, Scotland). All other chemicals including the buffer phosphate salts, maltodextrins and sodium chloride were obtained from Sigma-Aldrich® (Gillingham, UK).
Preparation of colistin sodium methanesulfonate (CMS) solution: A suspension of 2% gum arabic and 30% maltodextrin was prepared in a 200 mM potassium phosphate buffer (pH 6.0) and stirred under heat (25°C). This was allowed to cool to 15°C. Different volumes (15-25 mL) of a stock CMS solution (2000 Units mL-1) that had been prepared in 200 mM potassium phosphate buffer (pH 6.0) were added to give a 100 mL feed solution with different final CMS concentrations 300, 400 and 500 Units mL-1. This entire procedure was repeated with 2% gum arabic and 30% sodium chloride.
Spray drying of solutions: The CMS feed solutions were spray dried with a Büchi Mini Spray Dryer B-290 (Büchi Labortechnik, Postfach, Switzerland). The feed solutions were passed through an atomizing two-fluid nozzle (0.7 mm in diameter) into the drying chamber via a peristaltic feed pump at a feed flow rate of 3 mL min-1 and gas flow of 40 m3h-1. The inlet temperature (Tin) was 105±5°C while the outlet temperature (Tout) was at 57±9°C. The spray-dried particles were collected by a cyclone and stored below 8°C in a refrigerator until further analysis.
Determination of particle size distribution: The volume particle size distribution of the powders was measured using a dynamic light scattering instrument (Malvern Mastersizer Scirocco 2000, Malvern Instruments Ltd, Worcs., UK) with an in-house Standard Operating Procedure (SOP) (refractive index: 1.52; measurement time: 10 s; dispersive air pressure: 6 bar). The particle size distribution was characterized by the mass median diameter d (0.5) which is the value that 50% of the particles are smaller than while the other 50% are larger than it.
Characterization of particle morphology: The shape and surface morphology of the particles were examined by scanning electron microscope (SEM) (JEOL, model JSM-6490LV, Oxford, UK). The acceleration voltage was 15 kV and 1500 X magnification was used for all the samples. Prior to scanning, SEM stubs (diameter 12 mm) were dabbed on a dispersion of each powder and sputter coated with gold in an atmosphere of argon using a Polaron Equipment Ltd SEM Coating Unit E5100 sputter coater (Qurorum Technologies Ltd, East Sussex, UK) at 50 mA for 30 sec.
Determination of colistin sodium methanesulphonate (CMS) activity: This was achieved by comparing the inhibition of growth of Escherichia coli NCIMB 8879 produced by a solution of the spray dried powders and known concentrations of CMS that was not spray dried. E. coli NCIMB 8879 was cultured in 200 mL Luria Broth (LB) at 37°C for 24 h at a shaking speed of 120 rpm. This was then sub-cultured in LB at 37°C for 24 h. Two Hundred microliter of this sub-culture was grown in LB to an Optical Density (OD) of 0.129 at 600 nm which is the OD at 600 nm of a 0.5 McFarland Standard.
Hundred microliter of the sub-culture that had been grown to an OD of 0.129 was then spread on a Dey-Engley Neutralizing Broth (DENB) agar. Sterile adsorbent paper discs (diameter 6 mm) were impregnated with the solutions of a series of standard CMS as well as the spray dried powders. The standard solutions and the powders were prepared in a 200 mM potassium phosphate buffer (pH 6.0). The standard solutions were diluted at concentrations of 4000, 3500, 2000, 1000, 500, 300, 200 and 100 Units mL-1 while 20 mg mL-1 of the powders were prepared.
Three sterile discs impregnated with each solution were then placed on each inoculated DENB agar and the Petri dishes (86 mmx14 mm) were kept at room temperature for 1 h to allow the antibiotic solution diffuse. Afterwards, the Petri dishes were inverted and incubated at 37°C for 24 h.
The diameters of the circular inhibition zones were measured (in mm) using vernier calipers (Clarke® Electronic Digital Caliper) and a standard calibration curve was constructed. The residual activity of the spray dried CMS was then determined from the standard curve.
Data analysis: One of the packages in Microsoft Office Excel 2007® (Analysis of variance (ANOVA) single factor at an alpha value of 0.5) was used to analyse the size of the microparticles of spray-dried CMS using gum arabic and maltodextrin or sodium chloride.
Figure 1 shows the standard curve that was constructed by measuring the diameter of the zone of inhibition of the growth of Escherichia coli NCIMB 8879 cultured on Dey-Engley Neutralizing Broth agar at 37°C by standard concentrations 200, 300, 500, 1000, 2000, 2500, 3000 and 4000 Units mL-1 of unprocessed CMS.
| Table 1: | Microbiological activity of (300, 400 and 500) Units mL-1 of colistin sodium methanesulfonate (CMS) spray dried using 2% gum arabic and 30% maltodextrin or 30% sodium chloride |
 | |
| Spray drying was performed at an inlet temperature (Tin) of 105±5°C and an outlet temperature (Tout) of 57±9°C. The indicator organism used for the assay was Escherichia coli NCIMB 8879. ND = Not Detectable. Two inhibition zones had diameters outside the calibration curve. Hence the values shown are extrapolations of the average diameter of the zones of inhibition | |
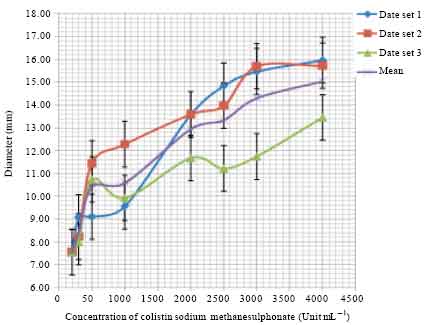 | |
| Fig. 1: | Standard calibration curve for the estimation of the bactericidal activity. Measurements were made in triplicate using a Vernier caliper (Clarke® Electronic Digital Caliper) with a precision of 0.01 mm |
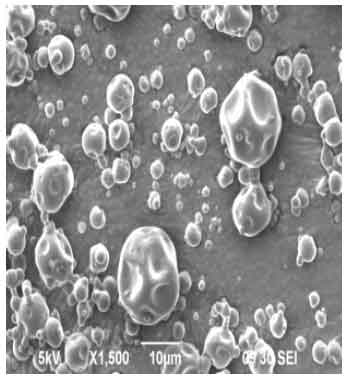 | |
| Fig. 2: | Scanning electron micrograph (SEM) of microparticles of 300 Units mL-1 colistin sodium methanesulphonate encapsulated in gum arabic and maltodextrin. The SEM was operated at 5 kV and secondary electron emission detected |
 | |
| Fig. 3: | Scanning electron micrograph (SEM) of microparticles of 400 Units mL-1 colistin sodium methanesulphonate encapsulated in gum arabic and maltodextrin. The SEM was operated at 5 kV and secondary electron emission detected |
 | |
| Fig. 4: | Scanning electron micrograph (SEM) of microparticles of 500 Units mL-1 colistin sodium methanesulphonate encapsulated in gum arabic and maltodextrin. The SEM was operated at 5 kV and secondary electron emission detected |
Table 1 shows the residual bactericidal activity of different concentrations 300, 400 and 500 Units mL-1 of spray-dried CMS on Escherichia coli NCIMB 8879. This shows that the 300 Units mL-1 CMS spray dried in gum arabic and maltodextrin did not show any antimicrobial activity. However, it clearly shows that encapsulation of 400 and 500 Units mL-1 CMS in gum arabic and maltodextrin resulted in residual bactericidal activities of 50% and 60%, respectively.
 | |
| Fig. 5: | Scanning electron micrograph (SEM) of microparticles of 300 Units mL-1 colistin sodium methanesulphonate encapsulated in gum arabic and sodium chloride. The SEM was operated at 5 kV and secondary electron emission detected |
 | |
| Fig. 6: | Scanning electron micrograph (SEM) of microparticles of 400 Units mL-1 colistin sodium methanesulphonate encapsulated in gum arabic and sodium chloride. The SEM was operated at 5 kV and secondary electron emission detected |
Figures 2-4 are Scanning Electron Micrographs (SEM) showing the shape and surface characteristics of the particles of the different concentrations 300, 400 and 500 Units mL-1 of Colistin Sodium Methanesulphonate (CMS) encapsulated in gum arabic and 30% maltodextrin. It is revealed that spray drying of CMS in gum arabic and maltodextrin resulted in microparticles that had smooth, non-uniform spherical surfaces with dents. On the other hand, Fig. 5-7 are SEMs of microparticles formed by spray drying different concentrations 300, 400 and 500 Units mL-1 of CMS in gum arabic and sodium chloride. The SEMs show microparticles with amorphous and irregular surface features. Figure 2-7 also contain information about the size of the particles.
 | |
| Fig. 7: | Scanning electron micrograph (SEM) of microparticles of 500 Units mL-1 colistin sodium methanesulphonate encapsulated in gum arabic and sodium chloride. The SEM was operated at 5 kV and secondary electron emission detected |
 | |
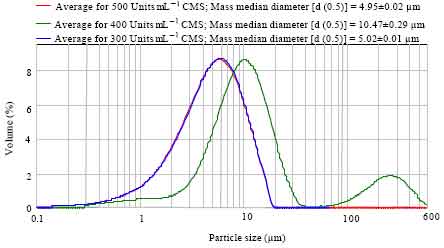
| Fig. 8: | Particle size distribution of 300, 400 and 500 Units mL-1 of spray dried colistin sodium methanesulphonate (CMS) in 2% gum arabic and 30% maltodextrin. Each curve is the average of three measurements of the same sample and is expressed as (Mean of mass median diameter±SEM). The value of d (0.5) signifies that 50% of the particles are smaller than this value while the other 50% are larger than it (Malvern Mastersizer Scirocco 2000 User Manual, Malvern Instruments Ltd, Worcs., UK) |
 | |
| Fig. 9: | Particle size distribution of 300, 400 and 500 Units mL-1 of spray dried colistin sodium methanesulphonate (CMS) in 2% gum arabic and 30% sodium chloride. Each curve is the average of three measurements of the same sample and is expressed as (Mean of mass median diameter±SEM). (Malvern Mastersizer Scirocco 2000 User Manual, Malvern Instruments Ltd, Worcs., UK) |
They show that the maximum size of the microparticles of CMS formed using gum arabic and maltodextrin or sodium chloride is 10 μm.
The additional information about the particle size distribution obtained from the Malvern Mastersizer Scirocco 2000 (Malvern Instruments Ltd, Worcs., UK) shows that 50% of the microparticles of CMS formed using gum arabic and maltodextrin are below 10 μm (Fig. 8). It was also observed that the concentration of CMS affected the size distribution of the microparticles formed using gum arabic and maltodextrin (Fig. 8).
On the other hand, Fig. 9 shows that spray drying of 300 and 400 Units mL-1 CMS in gum arabic and sodium chloride resulted in a particle size distribution of ~5 μm whereas a value of ~10 μm was obtained with the 400 Units mL-1 CMS solution.
The suitability of gum arabic for encapsulation of flavours by spray drying is an established norm in the food industry (Krishnan et al., 2005). This technology can be exploited to encapsulate biologically active compounds since carbohydrates are natural polymers that are biocompatible, non-toxic and are readily available (Sham et al., 2004). In this study, Colistin Sodium Methanesulfonate (CMS) was successfully encapsulated in gum arabic and maltodextrin with retention of biological activity.
It was observed that there was no inhibition of growth of the indicator organism Escherichia coli NCIMB 8879-when 100 Units mL-1 of the standard unprocessed CMS was used but there was inhibition with the 200 Units mL-1 CMS solution. Therefore, the average of 100 and 200 Units mL-1 is the Minimum Inhibitory Concentration (MIC). In other words, in this study, the MIC for CMS was estimated to be 150 Units mL-1 and is defined as the minimum concentration of CMS that is required to inhibit the visible growth of the indicator microorganism. This information was necessary for the choice of the range of standard concentrations needed to construct the standard calibration curve shown in Fig. 1.
Table 1 shows that the microparticles produced by spray drying the different concentrations of CMS in 2% gum arabic and 30% sodium chloride did not have any detectable microbiological activity against the indicator organism-Escherichia coli NCIMB 8879. The lack of activity could be due to the high ionic strength of the 30% sodium chloride. This is in agreement with a study that reported a six-fold decrease in the activity of gentamicin and tobramycin as a result of increasing the salt concentration of a medium from 22 to 174 mmol L-1 (Amsterdam, 2005). It is very likely that re-dissolution of the spray-dried CMS in the 200 mM phosphate buffer (pH 6.0) prior to the microbiological assay dramatically increases the osmolarity of the solution. The absence of activity could also be due to very poor encapsulation, if any, as shown by the amorphous SEM photographs in Fig. 5-7.
Conversely, Table 1 clearly shows that encapsulation of 400 and 500 Units mL-1 CMS in gum arabic and maltodextrin resulted in residual bactericidal activities of 50 and 60%, respectively which strongly suggests that maltodextrin could play a protective role compared to the lack of activity in the presence of sodium chloride. This finding appears to support the results of a work where spray-dried powders of ciprofloxacin and doxycycline, in the absence of carriers, retained above 90% of their antimicrobial activity (Adi et al., 2008). The difference in the retention of activity might be due to some unfavourable interactions between the positively charged CMS and the carriers/excipients (i.e., the mainly gum arabic and maltodextrin) that is absent in the spray drying of ciprofloxacin and doxycycline.
Table 1 also shows that the 300 Units mL-1 CMS spray dried in gum arabic and maltodextrin did not show any antimicrobial activity. It is possible that CMS was still encapsulated as shown by the spherical particles in Fig. 2 but the residual amount of the antibiotic after spray drying might be below the Minimum Inhibitory Concencration (MIC) which was estimated, in this study, to be 150 Units mL-1 and thus account for the lack of antibacterial activity. A simple and sensitive High Performance Liquid Chromatography (HPLC) method used in a previous study to assay for CMS in plasma and urine could be adapted to ascertain the amount of residual CMS in this spray dried sample (Li et al., 2002).
Table 1 also suggests that there might be an increase in the residual activity of spray-dried CMS in the presence of gum arabic and maltodextrin as the concentration of CMS in the spray drying feed solution increases. Higher concentrations of CMS in the feed solution will have to be spray-dried to confirm this.
This study also revealed that the microparticles generated by spray drying CMS in gum arabic and maltodextrin had smooth, non-uniform spherical surfaces with dents (Fig. 2-4). This is in agreement with a similar successful encapsulation process that involved spray drying cumin oleoresin in a blend of gum arabic, maltodextrin and modified starch (Kanakdande et al., 2007). On the other hand, spray drying of the different concentrations 300, 400 and 500 Units mL-1 of CMS in gum arabic and sodium chloride resulted in particles with amorphous and irregular surface features as shown in Fig. 5-7. This is characteristic of a spray drying process that did not result in encapsulation (Maa et al., 1997).
The additional information about the particle size distribution obtained from the Malvern Mastersizer Scirocco 2000 (Malvern Instruments Ltd, Worcs., UK) shows that 50% of the microparticles of CMS formed using gum arabic and maltodextrin are below 10 μm (Fig. 8). It was also observed that the concentration of CMS affected the size distribution of the microparticles formed using gum arabic and maltodextrin (Fig. 8). There was a progressive increase in the particle size distribution (from 7.22 to 9.52 μm) as the concentration of CMS increased from 300 to 500 Units mL-1 (Fig. 8). This could be due to an increased encapsulation efficiency which might be as a result of increased interaction of higher concentrations of CMS with the gum arabic and maltodextrin. This trend might account for the observed residual antimicrobial activity of spray-dried CMS. This finding contradicts the results of a study where a tetracycline-based antibiotic, doxycycline (DXY) was encapsulated in poly (lactide-co-glycolide) by spray drying (Patel et al., 2008). This difference may be due to the different interactions of each antibiotic with the wall-forming polymer. The hydrophilic character of DXY might reduce its interaction with the hydrophobic wall. Further studies will be required to ascertain the strength of the interaction between the cationic CMS and the water-soluble gum arabic and maltodextrin.
On the other hand, a particle size distribution of ~5 μm was obtained when 300 and 500 Units mL-1 CMS was spray dried in gum arabic and sodium chloride (Fig. 9). The apparently large mass median diameter of the 400 Units mL-1 CMS solution might be due to agglomeration of the particles that might have been caused by the presence of moisture in the powder. This is in agreement with a study where agglomeration caused an increase in the mean diameter of microparticles of esters spray dried in maltodextrin and modified starch (Buffo et al., 2002).
The series of experiments reported in this study strongly supports the claim in the literature that maltodextrin might interact favourably with the core of a spray-dried microparticle to prevent it from the stresses that might occur during spray drying. These experiments with maltodextrins also provide support for the current shift from the use of synthetic polymers to natural polymers as excipients in both the food and pharmaceutical industries.
Further studies will be required to optimize the spray drying parameters such as feed solution concentrations and feed solution flow rate with a view to improving the residual activity of the antibiotic after spray drying. It will also be crucial to determine the drug release profile of the spray-dried microparticles by determining the bactericidal activity over a period of time so as to evaluate the suitability of the encapsulation process for slow continuous release. This will also give an idea of the shelf life of the spray-dried particles. The study may also be extended to determine the most appropriate storage conditions for the spray-dried particles.
The findings of this work suggest that spray drying of Colistin Sodium Methanesulphonate (CMS) in gum arabic and maltodextrin appeared to have been successful as it resulted in the production of spherical microparticles with a size distribution of <10 μm which might satisfy a key requirement for deposition in the deeper parts of the lungs. In addition, a minimum of 50% of the antibacterial activity of CMS was retained after spray drying in gum arabic and maltodextrin.
I am immensely grateful to Dr. Steve Minter and other staff of Eminate Ltd., Biocity, Nottingham, UK for providing me with the reagents and laboratory where this work was carried out. I am also indebted to the Commonwealth Scholarships Commission, UK for providing the funds that enabled me go to the UK for further studies part of which included this work.