Research Article
Solubilization of Tricalcium Phosphate by Fungus Aspergillus niger at Different Carbon Source and Salinity
Department of Soil Science and Agricultural Sciences, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, India
Jay Prakash Verma
Department of Botany, MMV, Banaras Hindu University, Varanasi-221005, Uttar Pradesh, India
Kavindra Nath Tiwari
Department of Botany, MMV, Banaras Hindu University, Varanasi-221005, Uttar Pradesh, India










Sanjay Joshi Reply
I tried it.How it is possible,enzyme activity ends at 10 days only after wards it shows decrease in OD.
Please clarify it.
JAY PRAKASH VERMA
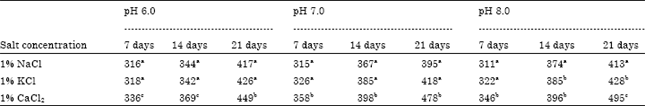
Sanjay ji, In our publication, enzyme activity will depend on the microbial physiology and their growth activities. In this publication, "solubilization of Tricalcium Phosphate by Fungus Aspergillus niger at Different Carbon Source and Salinity" I have study the solubilization capability, with different ph variation and carbon sources at 7, 14 and 21 days incubation. In this paper, Aspergillus grow continuously in the medium till the carbon source is available for growth.I am not determinate the enzyme activities, but I am sure, the enzyme activities enhance at time of fungal growth and large biomass production. when the nutrient is decreases then the fungal growth will also decrease, by which the enezyme activities also decreases. Enzyme activities will also depend on the carbon source and specific ph of the medium for growth of the microbial cell.