Research Article
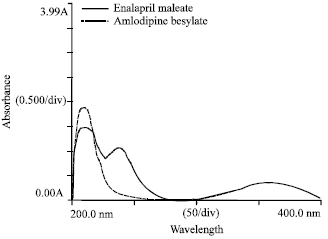
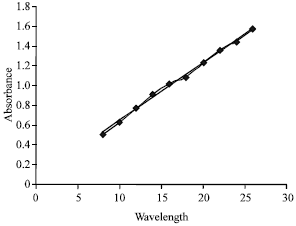
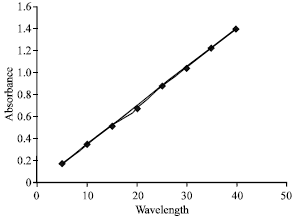
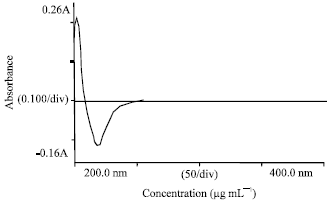
Development and Validation of Simultaneous Estimation of Enalapril Maleate and Amlodipine Besylate in Combined Dosage Forms
Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur (C.G.) 492 010, India
Shailendra Saraf
Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur (C.G.) 492 010, India
Swarnlata Saraf
Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur (C.G.) 492 010, India