Research Article
Statistical Model for the Evaluation of Corrosion Behaviour of Al-Sn Binary Alloy Systems
Department of Industrial Physics, Faculty of Applied Natural Sciences, Ebonyi State University, P.M.B. 053, Abakaliki, Ebonyi State, Nigeria
Material degradation, a consequence of physiomechanical interaction between materials mostly metals and its alloys with diverse environments have been a worldwide problem (Ekuma et al., 2007a). This is due basically to the complexity of the corrosion process which may be better appreciated by recognizing that many variables are involved: Environmental, electrochemical and metallurgical history of the material. For example, anodic reactions and oxidation rate, cathodic reaction and rate of reduction, corrosion inhibition, polarization or retardation; passivity, effect of oxidizers, effect of velocity, temperature, corrosive concentration, galvanic coupling and metallurgical structure all influence the type and rate of the corrosion process (Ekuma et al., 2007b).
Just as stated earlier, corrosion process is a complicated phenomenon and as such can be investigated to a very great extent by applying its real nature, which is mainly statistical and can be of various types due to the many forms of corrosion. The estimation of corrosion process using statistical techniques has been predicted for decades but its widespread application has been hampered largely because, the use of statistics requires professional knowledge and no reference standard(s) exist. For instance, microscopic variations in a surface tend to cause different forms of corrosion and also variations in the corrosion rate over a pit area. In these areas the simple assumption that corrosion rate is uniform across an area is unlikely to be accurate and sample thickness measurements are unlikely to be a good representation of the whole component but, the rapid self healing of the protective oxide layer on aluminium is of great advantage with regards to many forms of corrosion thus, the yardstick for its application in corrosion analysis (Idenyi et al., 2006).
Over the years, several researchers in their quest to formulate adequate model for the prediction of the service life of materials in diverse environments especially as regards answering such questions as the durability of metallic structures, determining the economic costs of damages associated with the degradation of materials (Feliu and Morcillo, 1993) and acquiring knowledge about the effect of environmental variables on corrosion kinetics (Ekuma, 2006) have developed many models.
Top most of these models are the deterministic and statistical models, which have been developed especially for the better understanding of the environments. Deterministic models are based on basic mathematical descriptions of atmospheric processes, in which effects (air pollution) are generated by causes (emissions). Examples of the deterministic models are Euler models (Zannetti, 1983) and Gaussian models (Zannetti, 1990). The statistical models are based on somewhat semi-empirical statistical relations between available data and measurements that do not necessarily reveal any relation between cause and effect. Basically, statistical models attempt to determine the underlying relationship between sets of input data (predictors) and targets (predictands). Examples of statistical models are regression analysis (Abdu-Wahab and Al-Alawi, 1996; Maliki, 2005), time series analysis (Hsu, 1992).
The aim of this study is to develop model equations using the regression analysis for the prediction of the performance of Al-Sn alloy systems in selected media environments (H2SO4, HCl and NaCl). The regression analysis (YCPR: Xt) will be used to develop a logarithmic model equation for the Corrosion Penetration Rate (CPR) as a function of time with the uttermost aim to fit different regression models developed using the SPSS 11.0 program and compare their performance with experimental data. It is envisaged that these analyses will be of uttermost importance in both material selection and in general corrosion design since corrosion processes in general is a complex statistical phenomenon. For proper comparison, the experimental results will be compared with the data generated using the developed model equations, which will be plotted and displayed for proper scrutiny. The data of Idenyi et al. (2004) was used for this analysis.
Figure 1-12 show the comparison plots of corrosion penetration rate as a function of time for the experimental data and model data for the various samples: Al-2.5% Sn and Al-4.5% Sn in various concentration media (0.5 and 1.0 M of H2SO4, HCl and NaCl, respectively).
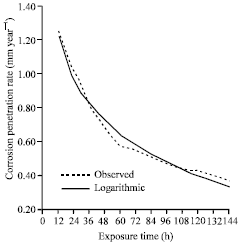 | |
| Fig. 1: | The corrosion penetration rate as a function of time for the experimental data and model data for sample A in 0.5 M H2SO4 media concentrations |
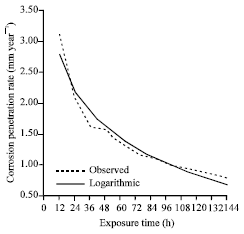 | |
| Fig. 2: | The corrosion penetration rate as a function of time for the experimental data and model data for sample A in 1.0 M H2SO4 media concentrations |
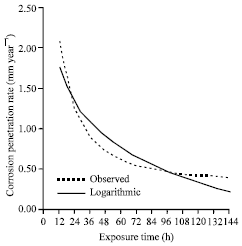 | |
| Fig. 3: | The corrosion penetration rate as a function of time for the experimental data and model data for sample A in 0.5 M HCl media concentrations |
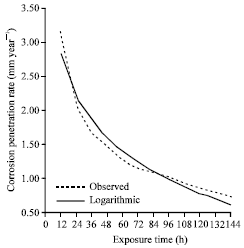 | |
| Fig. 4: | The corrosion rate as a function of time for the experimental data and model data for sample A in 1.0 M HCl media concentrations |
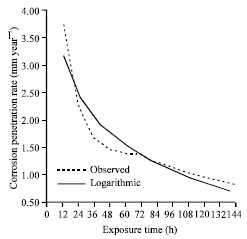 | |
| Fig. 5: | The corrosion penetration rate as a function of time for the experimental data and model data for sample A in 0.5 M NaCl media concentrations |
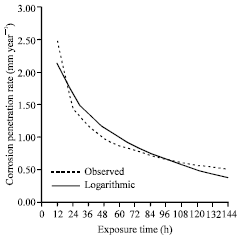 | |
| Fig. 6: | The corrosion penetration rate as a function of time for the experimental data and model data for sample A in 1.0 M NaCl media concentrations |
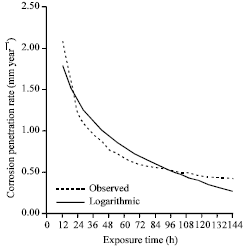 | |
| Fig. 7: | The corrosion penetration rate as a function of time for the experimental data and model data for sample B in 0.5 M H2SO4 media concentrations |
 | |
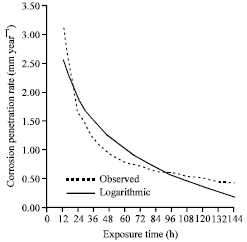
| Fig. 8: | The corrosion penetration rate as a function of time for the experimental data and model data for sample B in 1.0 M H2SO4 media concentrations |
 | |
| Fig. 9: | The corrosion penetration rate as a function of time for the experimental data and model data for sample B in 0.5 M HCl media concentrations |
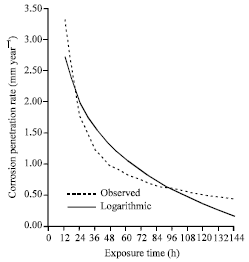 | |
| Fig. 10: | The corrosion penetration rate as a function of time for the experimental data and model data for sample B in 1.0 M HCl media concentrations |
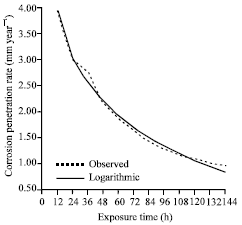 | |
| Fig. 11: | The corrosion penetration rate as a function of time for the experimental data and model data for sample B in. 0.5 M NaCl media concentrations |
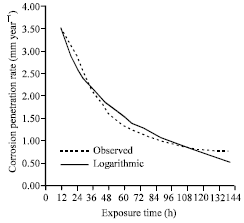 | |
| Fig. 12: | The corrosion penetration rate as a function of time for the experimental data and model data for sample B in.1.0 M NaCl media concentrations |
ANALYSIS OF RESULT
The regression analysis (Kleinbaum et al., 1998; Spiegel et al., 1999) of the corrosion penetration rate as a function of time was carried out. From the initial knowledge of the trend of the corrosion rate profile, the logarithmic regression analysis for the various environments was determined. In discussing the trend of the developed model equations, it will be looked into based on the alloy composition that is: Al-2.5% Sn and Al-4.5% Sn, respectively.
Analysis of Corrosion Rate for Al-2.5% SN Tetraoxosulphate (vi) Acid Environments
The tetraoxosulphate (vi) environment is of two concentrations: the 0.5 and 1.0 M. To investigate the corrosion trend in these environments, their regression models were developed.
For the Al-2.5% Sn binary alloy system in 0.5M H2SO4, the model equation is:
| (1) |
with coefficient of correlation (r-value) of 0.99022; coefficient of determination (R2-value) of 0.98053 and standard error of 0.04046. Where YCPR is the corrosion penetration rate in mm/ year and Xt is the exposure time in hours.
In order to check for the reliability of the developed model Eq. 1, we have to compare the modeled values with experimental data. To achieve this, we generated their corresponding values for the various time of exposure. As it can be seen from Fig. 1, the modeled values plotted with the experimental data have good correlation. This can further be clarified with the high positive coefficient of correlation: R = 0.99022. The coefficient of determinant shows that 98.05% of the variation of time depends entirely on the exposure time where only 1.95% is due to some other factors.
For the Al-2.5% Sn duplex alloy system in 1.0M H2SO4 concentration media, the developed model equation is:
| (2) |
It has coefficient of correlation, R = 0.97819; coefficient of determination which measures the total variation in CPR that has been explained by exposure time as R2 = 0.95686 and standard error, SE = 0.14386. Clearly, R2 ≈ 1 and the conclusion that can be drawn from this is that the model fit developed is precise and will be a good predicator of the behaviour of the alloy of aluminium (in this case Al-2.5% Sn) in the environment being studied. This can further be verified from Fig. 2 which shows good fit between the modeled values and experimental data.
Hydrochloric Acid Environments
Just like the tetraoxosulphate (vi) acid environments, the hydrochloric acid environments have two concentrations: 0.5 and 1.0 M concentration of HCl.
For the Al-2.5% Sn alloy system in 0.5M HCl media concentration, the model equation is:
| (3) |
where symbols retain their usual meaning. It has coefficient of correlation, r = 0.94935; coefficient of determination, R2 = 0.90126 and standard error, SE = 0.16231.
To check the validity of the developed equation, the experimental data is again compared with the modeled values (Fig. 3). It can be inferred (Fig. 3) that the modeled values fitted well with the experimental data. This is significantly supported by the high positive coefficient of correlation observed. It can further be pointed out that 90.13% of the total variation in CPR is accounted for by the variation in exposure time while only 9.87% is attributed to the influence of other factors not explained by the model equation.
The Al-2.5% Sn alloy system in 1.0M HCl media concentration has a model equation of the form:
| (4) |
with r-value of 0.97840, R2-value of 0.95726 and SE-value of 0.14823.
Using the model Eq. 4, the corrosion penetration rate values for the corresponding exposure time where generated and used for the comparison plot of the experimental values and modeled values (Fig. 4). From Fig. 4, it can be concluded that there is a good fit between the experimental and modeled values. This can further be verified from the high positive coefficient of correlation observed. It can also be seen that the coefficient of determination is ≈ 1 making the model equation a good predicator for the evaluation of the corrosion behavior of the alloy in the environment being studied.
Brine (NaCl) Environments
The corrosion behaviour of many alloys of aluminium has been investigated in diverse brine concentrations (Idenyi et al., 2006; Ekuma and Idenyi, 2006). In all, it was found to be influenced by the media concentration.
For the Al-2.5% Sn alloy system in 0.5M NaCl media concentration, the model equation obtained is:
| (5) |
It has correlation coefficient of 0.95146, coefficient of determination value of 0.90527 and standard error value 0.26380.
From Fig. 5, it can be inferred that the developed model Eq. 5 can be used as a good predicator of the alloy behaviour in the studied environment as the modeled values fitted well with the experimental data. This can further be clarified from the high positive coefficient of correlation that abound. Again, the analysis shows that 90.53% of the variation of the corrosion penetration rate is accounted for by the corresponding variation in time while only 9.47% is caused other parameters. This is known as the coefficient of alienation.
Also for the Al-2.5% Sn binary alloy system in 1.0 M NaCl media concentration, the model equation obtained is:
| (6) |
with R-value of 0.95772, R2-value of 0.91722 and SE-value of 0.16983.
Next, we check the correctness of the model equation developed. To achieve this, the model equation is used to generate values and a comparison plot of the experimental data and modeled data plotted (Fig. 6). Form Fig. 6, it can be inferred that there is a good fit between the experimental-and the modeled-data. This is overwhelmingly confirmed by the high positive coefficient of correlation (r = 0.95772) observed and further elucidated by the high positive coefficient of determinant (r = 0.91722).
Analysis of Corrosion Rate for Al-4.5% SN Tetraoxosulphate (vi) Acid Environments
As stated earlier, 0.5 and 1.0 M H2SO4 concentrations were used. Following the former section, we develop the model equations for the two media concentrations.
For the Al-4.5% Sn duplex alloy system in 0.5 M H2SO4, the model equation obtained is:
| (7) |
where symbols again retain their usual meaning. It has coefficient of correlation of 0.95662; coefficient of determination, r2 = 0.91512 and standard error, SE = 0.14725.
In order to establish the correctness of the developed model equation, we have to compare it with the experimental data. Hence we generated from (7) modeled values which for proper comparison was plotted with the experimental data (Fig. 7). From the plot, it can be concluded that there is significance correlation between the experimental-and modeled-data consequent upon its high positive coefficient of correlation. This can further be clarified from the high coefficient of determination (R2 = 0.91512) which shows that 91.51% of the total variation in CPR is attributed to corresponding variation in exposure time.
Also the Al-4.5% Sn binary alloy system in 1.0M H2SO4 has its own model equation which was obtained as:
| (8) |
It has coefficient of correlation of 0.99519, coefficient of determination of 0.99040 and standard error of 0.09029.
This uttermostly is a good fit as can be inferred from the high positive correlation coefficient and most especially from the high coefficient of determination which is approximately equal to unity. To further clarify this claim, the comparison plot (Fig. 8) was plotted from the generated modeled values.
Hydrochloric Acid Environments
Model equations were also developed for the two concentrations of HCl: 0.5 and 1.0 M habouring the duplex Al-4.5% Sn.
For the Al-4.5% Sn binary alloy system in 0.5M HCl, the model equation obtained is:
| (9) |
It has R-value = 0.93906, R2 value = 0.88183 and SE value = 0.27579.
In line with the aim of this research, we again compare the model parameters with the experimental data. It can easily be seen that the model fitted well with observed data (Fig. 9). The validity of the model is overwhelmingly confirmed by the high positive coefficient of correlation consequent upon high positive coefficient of determination which shows that 88.18% of the total variation in CPR is accounted for by the variation in exposure time.
Again, we obtain the model equation for Al-4.5% Sn binary alloy system in 1.0 M HCl as:
| (10) |
with coefficient of correlation as 0.94087, coefficient of determination of 0.88524 and standard error of 0.29312.
Sequel to the general format of this research, the experimental data is compared with the modeled data. To do this, the model Eq. 10 was used to generate corresponding corrosion penetration rate values for the whole exposure time. This was plotted with the experimental data (Fig. 10) for proper comparison. From Fig. 10, it can be inferred that the comparison plot shows that the model is in good agreement with the experimental data which can further be verified from the high positive coefficient of correlation.
Brine (NaCl) Environments
Model equations are also developed for the two concentrations of brine being investigated.
For the Al-4.5% Sn duplex alloy matrix in 0.5 M NaCl concentration media, the model equation is:
| (11) |
with R-value of 0.99582, R2-value of 0.99165 and SE of 0.09088.
A cursory look at the comparison plot (Fig. 11) generated for the modeled values using Eq. 11, it can be inferred that there is significant correlation between the experimental data and modeled data. This is further confirmed by the high positive correlation coefficient that is observed. Further scrutiny shows that 99.17% of the total variation in the CPR is explained by the variation in exposure time while only 0.83% is due to other factors. The implication of this is that the model equation can be used with high precision (SE = 0.09088) for the evaluation of the corrosion trend in 0.5M NaCl habouring Al-4.5% Sn duplex alloy system.
Also for the Al-4.5% Sn duplex alloy matrix in 1.0M NaCl media, the model equation is:
| (12) |
It has coefficient of correlation, r = 0.98574; coefficient of determination, R2 = 0.97168 and standard error, SE = 0.16137.
From the model parameters, it can be concluded that the model Eq. 12 is in good standing with the experimental data as can be seen from the high positive coefficient of correlation and markedly positive coefficient of determination.
From the model Eq. (1-12) developed and the statistical parameters obtained for each case, it is evident that all the model equations are good predicators of the corrosion susceptibility of the studied binary alloy systems in the various media being considered. The high positive coefficient of correlation is consistent for all the cases and also, the high coefficient of determination which for all cases is approximately unity with uttermostly low standard error further confirms this assertion. The implication of this singular observation is that all the developed model equations are in agreement with the experimental data. This can further be seen from the comparison plots (Fig. 1-12). However, this study in itself is not conclusive and as such recommended that a more elaborate work for other alloy compositions be carried out at expanded time scale to really determine the limit of these models.
I am most grateful to God for His kindness and love. Also to Dr. Maliki for useful discussion and Idenyi, N.E. for all his encouragement and permission to use their data.