Research Article
Roles of Alumina and Magnesia on the Formation of SFCA in Iron Ore Sinters
Department of Metallurgical and Materials Engineering, Obafemi Awolowo University, Ile-Ife, Nigeria
It has already been established (Ashan et al., 1983; Mulvaney, 1987; Nicola et al., 2004; Yang and Davis, 2005; Yang and Matthews, 1997) that the main matrix phase binding iron oxide grains in super fluxed sinters is a complex calcium ferrite known as SFCA (Silico-ferrite of calcium and aluminum). This phase has also been shown to have variable composition which is essentially iron oxide with a minimum of 3 wt% A12O3.
Proposals have been put forward as regards the mechanisms of formation of SFCA and these are undoubtedly influenced by the experimental methods adopted leading to some discrepancies as noted in an earlier work (Egundebi and Whiteman, 1989). Though A12O3 has been confirmed to be essential for the formation of this phase and MgO to inhibit its formation, there is still no general agreement on how A12O3 in particular interacts with other components such as Fe2O3, CaO and SiO2 in the mix to produce the complex phase. The main question is is A12O3 necessary for the presence of this phase or is it present in its analysis as a consequence of its being present in the raw mix and how does it favour the evolution of this phase?
The conclusions of Ashan et al. (1983) and Mulvaney (1987) that a critical level of A12O3 (3%) is necessary to stabilize SFCA in sinters arose from their analysis of phases in plant sinters. Their conclusions did not however take into consideration whether a phase with the same morphology would still have been produced if A12O3 were absent from the raw mix. Matsuno (1979) had earlier assumed that basicity was the only factor affecting the structure produced in sinters on cooling and that SFCA was essentially CaO.2Fe2O3 and that the effects of elements such as aluminum on the final structure are not important.
The roles of A12O3 have been predicted from composition of SFCA itself. Proposed mechanisms vary between the role of A12O3 as the initiator of SFCA formation and stabilizer once it has formed. The stabilizing effect of A12O3 has been reported by Yamaoka et al. (1974). It is considered that A12O3 initiates the formation of SFCA by reacting with Fe2O3 (with or without CaO) to form stable reactive intermediate compound. Mazanek and Jasienska (1964) proposed a similar mechanism for the formation of SFCA. Two alternatives are reported to be possible for the initial reaction in SFCA formation: either A12O3 reacts with Fe2O3 to give Al substituted hematite and the product then reacts with CaO.Fe2O3 and SiO2 to form substitute hemicalcium ferrite or SFCA; or A12O3 reacts with CaO to form CaO.A12O3 which dissolves in CaO.Fe2O3 to form a solid solution (Dawson et al., 1985). Inoue and Ikeda (1989) proposed that the initial reaction produces CaO.3(A1.Fe)2O3 which dissolves CaO.SiO2 to form SFCA but Dawson et al. (1985) found this was not the case in their investigations. Their results show that initial product is not CaO.3(A12Fe)2O3 but CaO.3(A12Fe)2O3 or monocalcium ferrite with Al substituted for some Fe in the hematite component.
In an attempt to explain the effect of alumina on the formation of SFCA, Mulvaney (1984) in his study constructed a pseudobinary system at two levels of Al2O3 and used this to consider the crystallization history of a model sinter of basicity equal to 2. He proposed that the presence of Al2O3 leads to a melt regime in which a peritectic reaction occurs between the initial magnetic and dicalcium silicate in the melt. But when alumima is absent, this reaction does not occur and only magnetite and dicalcium silicate are observed. What his work seems to suggest is that the role of Al2O3 in its influence on the formation of SFCA is in the melt rather than in initial reaction products, even though there is no way of his knowing of any initial reaction products since he was working on final sinter pieces. It is also clear from Mulvaney’s work that A12O3 enters the lattice of SFCA due to its being present in the melt. This mechanism supposes that SFCA forms by Fe2O3 and Al2O3 sandwiching themselves between rings of 2CaO.SiO2. This mechanism might not be wrong when it is considered that the Ca/Si ratio in 2CaO.SiO2 is the same as that reported in sinters.
If it is accepted that the effect of Al2O3 on the precipitation of SFCA occurs in the melt, then the earlier suggestion of Egundebi and Whiteman (1989) that it is the property of the melt that is important in the precipitation of SFCA becomes relevant. The property of the melt that is thought to be the most relevant is viscosity and a high viscosity melt is thought to favour the formation of SFCA. This allows an explanation of the role of Al2O3 in the melt since alumina is known to increase the visocity of alumino-silicate melts. The reason why MgO does not favour the formation of SFCA also becomes clear since MgO is also known to lower the viscosity of alumino-silicate melts.
The objective of this study is to asses the proper role of Al2O3 and MgO on the evolution of phases in sinters especially SFCA and therefrom establish a mechanism for the formation of the phases.
A fine iron ore (from Mt. Wright, Canada) consisting of about 94% hematite (the rest being SiO2 and Al2O3) was used. The ore was ground and sieved and -75 µm size fraction was used in the experiments. A commercial grade limestone containing about 7% SiO2 was used and this was also ground to the same size fraction as the ore. Mixtures of limestone and ore in the desired proportions were thoroughly mixed using mortar and pestle. For the experiments, a 80/20 ore/limestone mixture (by weight was used and the analysis of the mix (wt%) is:
75.7%Fe2O3, 5.4%SiO2 0.35% Al2O3 and 10.42%CaO (18.46%CaCO3) Basicity = 1.93 |
The experiments were carried out in a horizontal tube furnace which can be heated up to 1400°C by resistance heating. Sintering was carried out at temperatures ranging from 1100 to 1300°C for different times (1-10 min).
For positive identification of the mineral phases, powder samples were subjected to X-ray diffraction examination in a Philips Diffractometer (using Co-Ka radiation, 30 mA, 40 KV and diffractometer speed of 1° 2?/min).
Solid samples were also mounted for optical and scanning microscopic examinations. The samples being porous were mounted in vacuum to suck in the cold mounting resin into the sample. They were polished using paraffin since some of the phases in the sinter are soluble in water. The LINK energy dispersive microanalyser attached to the PSEM 500 electron microscope was used to obtain chemical analysis of the phases to supplement X-ray diffraction identification. The LINK energy dispersive X-ray microanalyser enables both qualitative (analysis using only the number of x-ray counts with no corrections) and quantitative analyses (with ZAF corrections-atomic number, absorption and fluorescence) to be carried out. Cobalt was used as standard and oxides for quantitative analysis. A filament voltage of 25 KV was used with a spot size of 64 nm to give sufficient number of counts (usually about 2000).
Quantitative determination of SFCA phase in the samples was carried out using X-ray diffraction and an internal standard method. A standard sample of SFCA (used for obtaining a calibration graph) was first prepared using pure samples of Fe2O3, CaO, SiO2 and Al2O3 in a Au/Pt/Rh crucible. Pure lime was used as the internal standard and its proportion in each of the experimental samples was 20%.
Effects of Different Levels of Al2O3
In order to assess the effect of different levels of alumina on evolution of phases, mixes containing 1 and 2% alumina were prepared. The natural ore which contains 0.35% Al2O3 was taken as base level. Sintering was carried out at 125°C for 4, 6 and 10 min sintering time at each level of alumina.
The results are best presented by comparison as is done in Fig. 1 which shows the micrographs of specimens prepared from mixes containing the 3 levels of alumina after 4, 6 and 10 min sintering times at temperature of 1250°C. The most notable observations from the results are:
| • | The sequence of formation of phases at the different levels of alumina is the same except that the rate at which SFCA appears and disappears are different. At base level, SFCA forms as early as 4 min (Fig. 1A) but the proportion is low compared to other levels. At 10 min, while SFCA is still present in the microstructure (Fig. 1C), there are significant amounts of magnetite and dicalcium silicate. |
| • | Addition of 1% Al2O3 alters the phase proportions. The micrograph at 4 min (Fig. 1D) contains almost 100% needle SFCA but at 6 min (Fig. 1E), the proportion of SFCA has significantly dropped while at 10 min (Fig. 1F), none is present. |
| • | At 2% Al2O3 the phase proportions are again different. The microstructure after 4 min (Fig. 1G) contains essentially needle SFCA up to 10 min (Fig. 1H and I). |
Using the amount of SFCA in the samples (by the use of X-ray diffraction internal standard method) to monitor progress of reaction, the result is as shown in Fig. 2. The result clearly shows that increasing the level of Al2O3 to 2% gives higher SFCA over a wider range than at lower Al2O3 levels.
Combined Effects of Al2O3 and Temperature
The experiments in the last section were carried out at 1250°C as indicated and from the results, it appears increasing alumina content might be more effective in obtaining more SFCA at long sintering times. From earlier results (Egundebi and Whiteman, 1989), activities at long sintering times can be equated to those at temperatures above 1250°C in terms of phases evolved. Therefore, in order to assess the effect of alumina when sintering is carried out at temperatures higher than 1250°C, samples containing different levels of alumina were sintered at 1300 and 1350°C for 4 min each.
 | |
| Fig. 1: | Optical x400 Photomicrographs of sinters produced at the levels of A1 and time shown. They consist essentially of SFCA, dicalcium silicate+glass and magnetite |
 | |
| Fig. 2: | SFCA (%) vs time for different levels of Alumina |
The results are as shown in Fig. 3, where SFCA content in the specimens has been plotted against temperature for different levels of alumina. The figure clearly shows that at temperatures above 1250°C (using the same sintering time), the amount of SFCA formed in the specimens increases with increase in % Al2O3. The effect of alumina is more significant at 1300°C where a 1% change Al2O3 in gives a 20% rise in SFCA content.
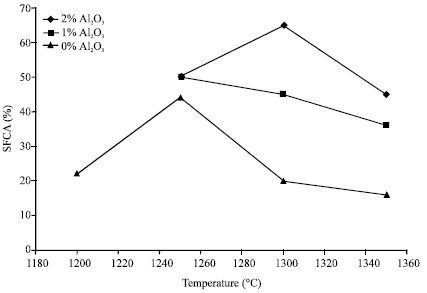 | |
| Fig. 3: | SFCA (%) vs temperature for different levels of Alumina |
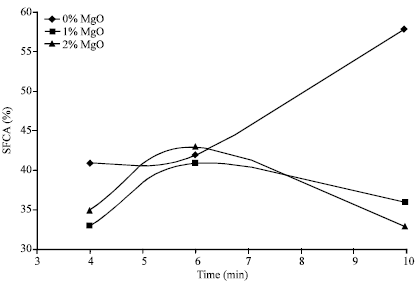 | |
| Fig. 4: | SFCA (%) vs time for different levels of MgO |
The effect of alumina can be said to be insignificant at 1250°C where the change in SFCA content is less than 5% for 1% change in alumina but as shown in Fig. 2, alumina effect might become less significant when sintering time exceeds 4 min. As temperature rises to 1350°C the beneficial effect of higher alumina content in terms of obtaining more SFCA diminishes again. This seems to indicate that as temperature rises, more alumina might be needed to obtain more SFCA.
Effect of Different Levels of MgO
The ore as received contains 0.01% MgO which is taken as zero. In order to assess the effect of MgO, two levels of magnesia (1 and 2%) were added to the ore. Samples were taken from the mixes and sintered at 1250°C for 4, 6 and 10 min at each level of MgO.
The results are as shown in Fig. 4 where the SFCA content in the samples have been used to monitor the progress of reactions. Figure 5A and D shows that inclusion of magnesia leads to delay in reaction at the start of sintering. Magnesia has an opposite effect to go magnesia situation where the SFCA content increase with time (Fig. 1: A-C).
 | |
| Fig. 5: | Optical x400 Photomicrographs of sinters produced at the levels of MgO and times shown. They consist of hematite (H), Magnetite (M), Magnesio-ferrite (MF), SFCA and dicalcium silicate+glass |
It also has an opposite effect to alumina, i.e., where alumina tends to become more effective as sintering time increases (Fig. 2). In terms of SFCA content, magnesia leads to lowering of SFCA content. Generally, magnesia does not appear to prevent the formation of SFCA, but the amount of SFCA obtained tends to become insignificant when compared to no magnesia situation as further shown in Fig. 5. Magnesia-ferrite tends to form in preferences to SFCA and a lot of glassy phase is also formed.
The effects of alumina and magnesia on the formation of SFCA in sinters as previously reported by other workers have been confirmed by this study. But the results do not support the previous conclusion that a level of alumina is necessary before SFCA can be obtained. MgO too does not prevent the formation of SFCA but leads to its being obtained in reduced quantities. The roles of Al2O3 and MgO derive from their influence upon melt properties. Two main things have emerged from these results:
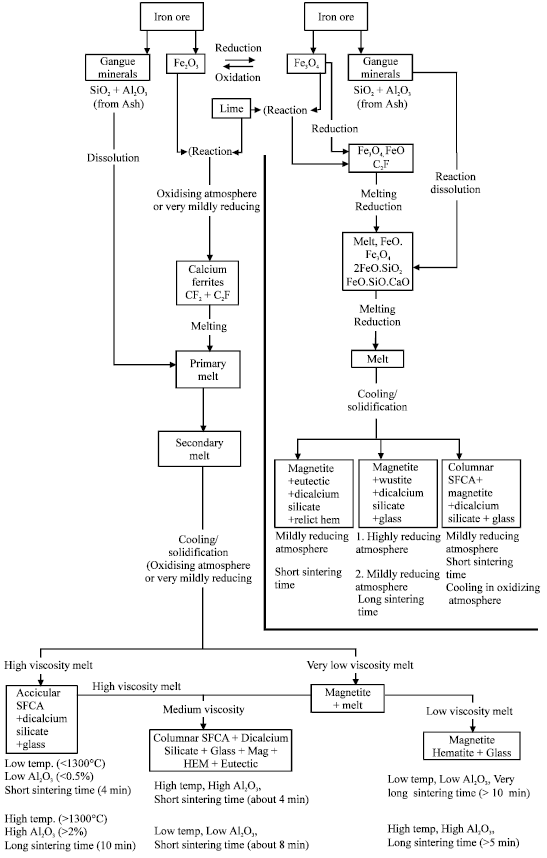 | |
| Fig. 6: | Schematic illustration of sintering mechanisms |
| • | Al2O3 allows SFCA to be obtained in sinters under the conditions where it would not have otherwise been obtained such as high temperature and low sintering times. Since SFCA is obtained from the experimental sintering mix containing only 0.35%, then SFCA can be obtained without alumina being present up to 3% as previously reported. |
| • | The fact that SFCA containing less than 1% Al2O3 is obtainable in sinters puts in doubt the role of alumina as a stabilizer of SFCA. It may happen that alumina only leads to a melt regime in which the formation of SFCA is made possible. The suggestion that SFCA is an intermediate phase which forms during sintering and disappear by melting either due to increase in temperature or sintering time may also not be true. This notion may have arisen from the experimental method adopted by some workers whereby a sinter is prepared, cooled down and reheated again to see which phases have disappeared. SFCA as an intermediate phase is not supported by the proposed mechanism of Mulvaney (1984) in which magnetite is proposed to be the intermediate phase. SFCA will or will not form depending on the state of the melt just before cooling and solidification starts. The role of alumina as stabilizer of SFCA may be valid if it is accepted that SFCA forms from the solid state but this has been proved doubtful from evidence provided in a previous study (Egundebi and Whiteman, 1989). |
Melt Viscosity and Roles of Al2O3 and MgO
It has been stated in a previous study (Egundebi and Whiteman, 1989) that the property of the melt that governs the crystallization of SFCA from the melt is viscosity and that high viscosity favours the formation of SFCA. The roles of Al2O3 and MgO consist of their effects on melt viscosity. Al2O3 increases melt viscosity while and MgO lowers it. Therefore, it appears that before SFCA can form, the silicate network has to be preserved and the melt must not be fluid.
Since SFCA is assumed to crystallize from the melt, then like all other crystals, it has to form by nucleation and growth process.
Sintering Mechanism
A sintering mechanism incorporating the idea of melt viscosity on the evolution of phases had been proposed earlier (Egundebi and Whiteman, 1989). This sintering mechanism is here modified to include the effects of Al2O3. This modified mechanism is shown in Fig. 6. Viscosity has made it possible to combine all the effects of composition, temperature and basicity on the formation of SFCA earlier proposed by other workers (Mulvaney, 1984; Ishikawa et al., 1983) and thereby resolve the discrepancies between their various proposals.
Implications of the Results for Sintering Technology
From the results of other workers (Dawson et al., 1985; Ishikawa et al., 1983), there seems to be a general agreement that it is sinter with an acicular calcium ferrite structure that is best for obtaining optimum results in the Blast Furnace. This acicular ferrite structure however derives from a sinter in which the SFCA has the needle morphology. The conditions under which the structure can be obtained have been stated in Fig. 6. However the first of the two sets of conditions should be the preferred one for reasons of savings in energy for sintering and operational ease in the Blast Furnace since a high alumina in sinter will lead to a high blast furnace slag viscosity.
The roles of Al2O3 and MgO on the formation of SFCA in sinters derives form their effects on melt viscosity. Al2O3 is a network former leading to a silicate melt of high viscosity while MgO is a network modifier leading to a silicate melt of low viscosity.
Al2O3 may not be necessary for the formation of SFCA if the conditions during sintering combine in such a way that the melt in which the SFCA precipitates out has a high viscosity,