Research Article
Evaluation of Influence of Process Variables on the Corrosion Performance of Hot-dip-galvanized Steel Sheets
Department of Metallurgical and Materials Engineering, Obafemi Awolowo University, Ile-Ife, Nigeria
Steel generally corrodes when freely exposed to air at between 0.05 mm per year to 0.125 mm per year (Lee, 1981). The higher rate of steel corrosion in more aggressive environment makes its protection inevitable especially when they are to be used in such environment. Many investigations conducted on the protection of steels from corrosion confirmed the superiority of Hot-Dip-Galvanizing method over others (Lee, 1981; Frihe and Hankel, 1996; Roebuk et al., 1984) thus it is the most widely used, the most effective and the most economical.
The current economic situation in developing countries makes it difficult to replace old galvanized products like buckets, garbage cans, water storage tanks and roofing sheets. It becomes necessary to improve the corrosion resistance of galvanized sheets so as to prolong their service life. Some investigators reported some process variables contributing to the corrosion performance of Holt-Dip-Galvanized sheets such as bath temperature, immersion duration and withdrawal speed (Hanna and Nassif, 1983; Burns and Bradley, 1967). Others added the effect of zinc bath additives like aluminum and antimony (Haarman and Horstmann, 1958; Vitikin, 1973; Pavlidis and Robert, 1976; Sinha and Miller, 1970).
The thickness of galvanized coatings has been said to determine its length of corrosion protection. It is now believed that other factors may play a role in ascertaining its true protective value (Mathewson, 1970; Arenas et al., 2006; Karakasch, 2001; Morimoto et al., 2003; Morishita et al., 2003; Vogt et al., 2001). Such factors might include the quality of coatings in terms of adhesion, uniformity and appearance.
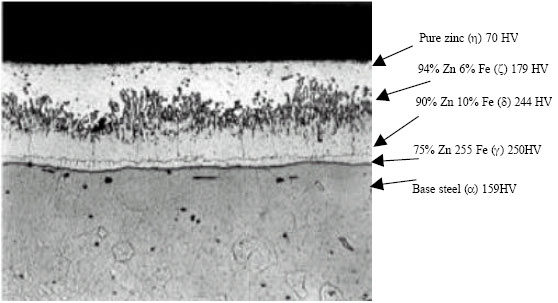 | |
| Fig. 1: | Photomicrograph of a galvanized steel coating cross-section shows a typical coating microstructure consisting of three alloy layers and a layer of pure metallic zinc. (HV stands for Vickers hardness) |
Galvanized coating comprises of outer pure zinc layer and several inner alloy layers of iron and zinc intermetallic phases, the layers becoming successively richer in iron with depth as shown in Fig. 1. (http://www.corrosiondoctors.org/MetalCoatings/ Galvanizing.htm). The role each of these layers plays in the overall corrosion performance of galvanized coating is not yet agreed upon.
In this study, the effect of galvanizing temperature, immersion duration and withdrawal speed on the corrosion resistance of Hot-Dip-Galvanized sheets will been determined. The optimum process variables and the evolved coating thickness will be established.
Laboratory galvanizing of steel sheets of gauges 18, 22 and 28 containing 0.15% C each and 0.09, 0.33 and 0.53% of silicon concentrations, respectively was carried out. A Carbolite vertical furnace, cylindrical steel crucible, 12V D.C electric motor whose speed is varied with a rheostat and a D.C generator were employed to accomplish the process. The three gauges of steel sheets were cut into 3x3 cm with 2 mm hole drilled at 2 mm to the edge of each sheet on the vertical central line. The steel sheets were decreased in ethanol for 30 min, pickled in 15.% HCI for 30 min, rinsed in running water for 10 min, fluxed in zinc ammonium chloride solution and dried on an hot plate at 100°C for a minute.
Zinc ingot (99.8% Zn) was charged into the steel crucible with one percent pure lead put in the bottom of the crucible. The three gauges of steel sheets were galvanized at 430, 450, 470 and 490°C for 30 sec, 1, 1.5 and 2 min at withdrawal speeds of 3, 4 , 5 and 6 m min-1. The galvanized steel plates were withdrawn into a clean area in an open space where they were rapidly cooled. They were later dipped in a dilute solution of potassium dichromate solution for passivation except for one set of gauge 22 sheets galvanized at 450°C for 1min and withdrawn at 4 m min-1. They were stored in desiccators for preservation before testing.
The stored sheets were tested for corrosion resistance in a salt spray chamber according to ASTM Standard B117 (2003). Weight losses were determined using *H51 Mettler chemical balance.
Coating Quality
The quality of the coatings produced was evaluated by their lustre, appearance and uniformity. The results are given in Table 1-8. The factors affecting the product quality are as follows.
Galvanizing Temperature
Steel sheets galvanized in pure zinc bath has bright appearance at 450°C, good uniformity (within 5 μm) without stains except gauge 18 sheet with dull gray coatings. The uniformity of coatings was poor (within 15 μm) at 430, 470 and 490°C for the gauges considered except for gauge 18 sheet with fair (within 10 μm) uniformity at 470. Galvanizing of gauge 28 sheet was almost impossible above 470°C owing to the heat effect on the thin sheet. This is shown in Fig. 2.
Immersion Time
Coating quality was found to decrease with increase in immersion time after sixty sec (Fig. 4). Non-uniform coatings with excessive thicknesses and stains were obtained after the optimum immersion time of 60 sec.
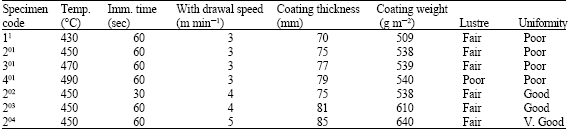
| Table 1: | Effect of galvanizing temperatures, immersion time and withdrawal speeds on gauge 28 steel sheet |
 | |
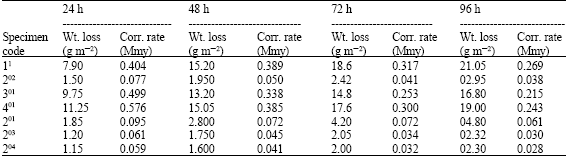
| Table 2: | Weight losses and corrosion rates of galvanized gauge 28 sheet after 24, 48, 72 and 96 h of 5% NaCl solution spray |
 | |
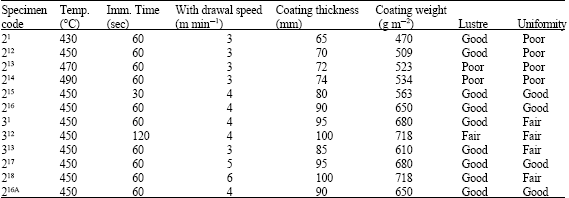
| Table 3: | Effect of galvanizing temperatures, immersion time and withdrawal speeds on gauge 22 steel sheet |
 | |
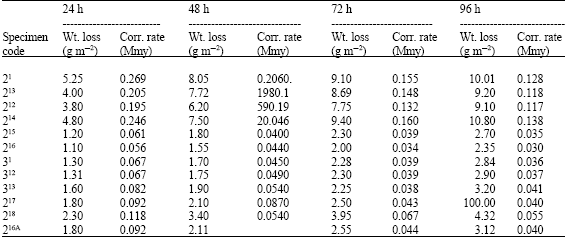
| Table 4: | Weight losses and corrosion rates of galvanized gauge 22 sheet after 24, 48, 72 and 96 h in spraying of 5% NaCl Solution |
 | |
| Table 5: | Effect of galvanizing temperatures, immersion time and withdrawal speeds on gauge 18 steel sheet |
 | |
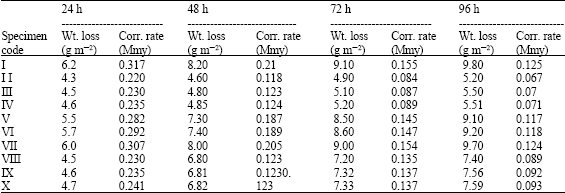
| Table 6: | Weight losses and corrosion rates of galvanized gauge 18 sheet after 24, 48, 72 and 96 h in spraying of 5% NaCl solution |
 | |
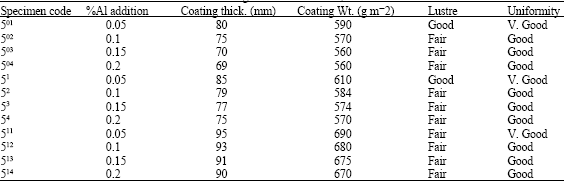
| Table 7: | Effect of Al addition to the bath on sheets galvanized at 450°C, for 60 sec withdrawn at 4 m min-1 |
 | |
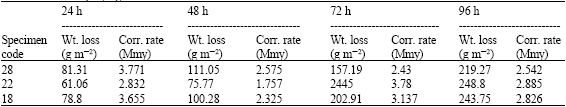
| Table 8: | Weight losses and corrosion rates of degreased, pickled and rinsed as received steel sheets after 24, 48, 72 and 96 h in spraying of 5% NaCl Solution |
 | |
Withdrawal Speed
Coating quality was found to depend on withdrawal speed in relation to sheet thickness. The gauge 28 sheet had the best coatings when withdrawn at 5 m min-1 with good appearance, shining lustre and very good uniform (within 2 m). However, gauge 22 sheet had the best coatings when withdrawn at 4 and 3 m min-1 for gauge 18 sheet. Gauge 28 sheet could not be withdrawn at 6 m min-1 owing to its lightweight and spillage of the molten zinc (Fig. 3).
Corrosion Performance of Coatings in Spraying of 5% NaCl Solution
Though the overall length of corrosion protection depends on the thickness of the coatings, the quality of the coatings was found to influence their corrosion resistance. The observed tested specimens showed evidence of uniform corrosion without spots or pittings.
Galvanizing Temperature
The corrosion resistance of the coatings was found to increase with increase in galvanizing temperature from 430 to 450°C bur decrease set in thereafter, for all gauges of galvanized sheets tested. Gauge 18 sheet had the highest resistance at 490°C because of its thickness (Fig. 2) 450°C was found to be optimum galvanizing temperature for the three gauges of sheets considered.
Withdrawal Speed
Corrosion resistance of the coatings was found to decrease with increase in withdrawal speed for gauge 18 coated sheet from 3 to 6 m min-1 at constant galvanizing temperature and immersion time. Gauge 22 and 28-coated sheet had their corrosion resistance increasing with increase in withdrawal speed having maxima at 4 and 6 m min-1, respectively. So the optimum withdrawal speeds were found to be 3 m min-1 for gauge 18, 4 m min-1 for gauge 22 and 5 m min-1 for gauge 28 (Fig. 3).
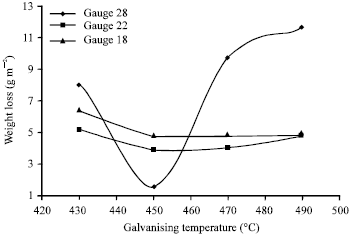 | |
| Fig. 2: | Weight loss vs galvanizing temperature |
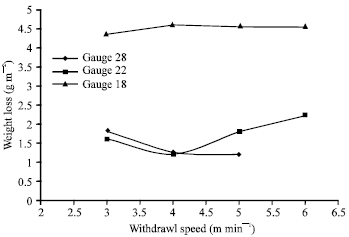 | |
| Fig. 3: | Weight loss vs withdrawal speed |
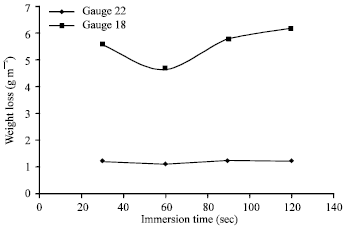 | |
| Fig. 4: | Weight loss vs immersion time |
Immersion Time
The corrosion resistance of galvanized sheets was found to increase with increase in immersion time till sixty sec (Fig. 4) thereafter decreasing effect was obtained at constant galvanizing temperature and withdrawal speed. Galvanizing of gauge 28 sheet could not be accomplished after sixty sec immersion time without heave stains.
Dipping in Potassium Dichromate Solution
Corrosion resistance of galvanized sheets was also enhanced by chromate dipping. This is due to passivation of the galvanized sheet as a result of the chromate layer.
Coating Thickness and Weight
The length of corrosion protection was found to depend on coating thickness and weight for uniformly coated sheets. The thicker the coating for uniformly distributed coatings and the bigger its weight, the longer the service life of the coating. The corrosion resistance however was found to depend on coating adhesion and uniformity rather than its weight or its thickness. The optimum coating thickness and their weight are found to be 85 μm (610 g m-2) for gauge 28,90 μm (650 g m-2) for gauge 22 and 10 μm (722 g m-2) for gauge 18. The corrosion resistance however was found to depend on coating adhesion and uniformity rather than on its weight or its thickness.
Silicon Concentration in Steel Sheet
The quality and corrosion resistance of galvanized sheets depends on the silicon concentration of the steel. In the range 0.05 to 0.12% Si steel fabrications galvanized conventionally receive excessively thick coatings with poor appearance and reduced adhesion. Thus gauge 18 sheet with 0.09% Si had dull and least corrosion resistant coatings.
An improvement in corrosion resistance of the galvanized sheets is enhanced by proper correlation of process variables with the sheet thickness hence the optimum galvanizing variables established are 450°C as galvanizing temperature, 60 sec as immersion time and withdrawal speeds of 3, 4 and 5 m min-1 for gauges 18, 22 and 28 sheets, respectively.
Though the length of corrosion protection is known to depend on the thickness of the coatings, the quality of the coatings in terms of lustre, appearance, adhesion and uniformity are found to determine their corrosion resistance. The established optimum coating thickness and weight are 85, 90 and 10 μm (722 g m-2) for gauges 28, 22 and 18 sheets, respectively.