Research Article
Cadmium Toxicity on Species of Bacillus and Pseudomonas During Growth on Crude Oil
Microbiology, University of Calabar, P.M.B. 1115, Calabar, Nigeria
S.P. Antai
Microbiology, University of Calabar, P.M.B. 1115, Calabar, Nigeria
One of the major drawbacks of microbial remediation of petroleum-polluted environments is the extremely slow rate at which biodegradation occurs (Cooney, 1984). A number of factors responsible, including both biological and environmental, have been identified, studied and reviewed (Atlas, 1981, 1992; Bartha and Atlas, 1977; Antai, 1990). Of primary importance are the nature and number of bioremediating microorganisms, as well as the presence of toxic metals at concentrations above trace amounts.
The growth of microorganisms is greatly affected by the chemical and physical nature of their surroundings (Prescott et al., 1999). The ability of microorganisms to grow and the overall rate at which they do so, markedly influence bioremediation processes. The influence of heavy metals on microbial processes has been reviewed (Baath, 1989). Heavy metals impact both the physiology and ecology of pollutant degrading microorganisms (Sandrin and Maier, 2003). By binding to enzymes involved in the general metabolism of biodegrading microorganisms, heavy metals influence negatively the effectiveness of bioremediation processes (Baldrian et al., 2000).
The research presented here concentrated on the effect of cadmium on the growth of Bacillus sp. and Pseudomonas sp., bacteria most frequently encountered in the oil-polluted environments in the Niger Delta region of Nigeria. Cadmium was chosen because it is reported to be an inducer of oxidative stress in microorganisms (Kachur et al., 1998).
The crude oil used for the study was obtained from Mobil Producing Nigeria Unlimited, Eket, Akwa Ibom State, Nigeria. Filter-sterilised oil was kept at room temperature (28±2°C) until required.
Reagents used in the study were of analytical grade and were obtained from the Departments of Chemistry and Biochemistry, University of Calabar, Nigeria. Concentrated stock solutions of cadmium were prepared from the chloride salts of the metal (CdCl2) according to the method described by Asuquo et al. (2004) and stored at 4°C until use (Koukal et al., 2003). Working concentrations of 1, 10, 100 and 1000 mg L-1 were obtained by serial dilutions of the concentrated stock solutions in sterile deionised distilled water (ddH2O).
The bacterial cultures used for the study were isolated from water and sediment samples obtained from Qua Iboe river estuary, in the Niger Delta region of Nigeria. The cultures were isolated on mineral salts medium (Zajic and Supplission, 1972) supplemented with Agar and pH adjusted to 7.2. The crude oil was supplied in vapour form from the lid of the plate (Thijsee and van der Linden, 1961) to provide carbon and energy source. The organisms were purified on Nutrient Agar (NA), maintained on slants containing media for their purification and identified according to the scheme of MacFaddin (1980).
Oil broth utilization test (Okpokwasili and Okorie, 1988) was used to screen the organisms for oil-utilization ability.
The influences of varying concentrations of cadmium on the growth of the isolates on crude oil/minerals salts medium were monitored turbidimetrically (Malakul et al., 1998). Study flasks were incubated at 30°C for 16 days. Determinations of Optical Density (OD) at 540 nm wavelength, pH and Total Viable Counts (TVC) were made on day 0, 1, 2, 3, 4, 8, 12 and 16. Using the total viable counts data, the number of generations, growth rate and generation times of the isolates were calculated.
The bacterial cultures were identified as species of Bacillus (from sediment sample) and Pseudomonas (from water sample), by the scheme of MacFaddin (1980). The organisms grew well in broth screen tubes, producing good turbidity as an indication of oil utilization.
In Fig. 1, the pH of the growth medium decreased from 7.20 to 5.91 at the end of the sampling period (16 days or 384 h), in the control flasks. Flasks containing various concentrations of cadmium similarly showed pH decrease, but not as in the control. For instance, in study flasks containing 1000 mg L-1 cadmium, the pH at 384 h incubation was 6.83 as against 7.20 at 0 h. pH changes in control flasks and those containing 1 mg L-1 cadmium were similar up to 192 h where their respective pH values were 6.32 and 6.42. Optical density (OD) readings and the total viable counts (TVC) increased as incubation period increased. In the control flasks, TVC data increased from 0.38x103 CFU mL-1 (0 h) to 2.23x1012 CFU mL-1 (384 h), with 0.026 and 1.286 OD readings, respectively. TVC data from cultures containing 1000 mg L-1 cadmium was 0.99x105 CFU mL-1 at 384 h. Values for cultures containing lower concentrations ranged from 106 to 1012 CFU mL-1. In Fig. 2, the pH dropped from 7.23 to 6.23 (at 384 h) in the control and 6.92 in flasks containing 1000 mg L-1 cadmium.
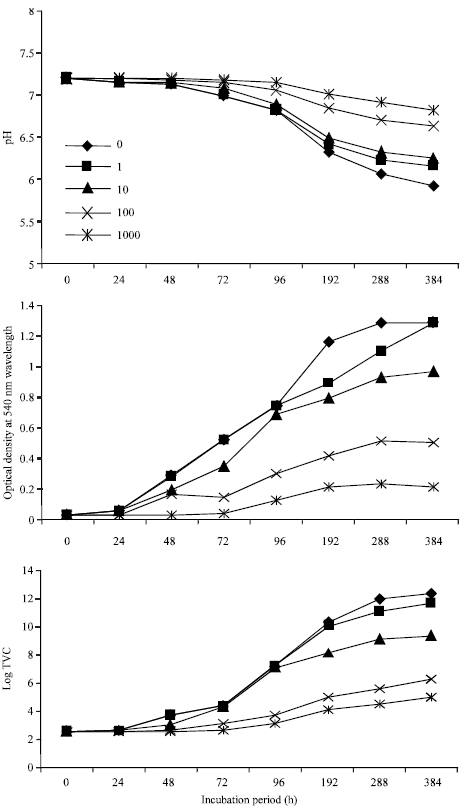 | |
| Fig. 1: | Influence of cadmium on the growth profile of Bacillus sp. on crude oil |
The pH, OD and TVC values in the control and cultures containing 1 mg L-1 cadmium were similar up to 288 h where both cultures had values of 6.28, 1.152 and 1.88x1011 CFU mL-1 for pH, OD and TVC, respectively.
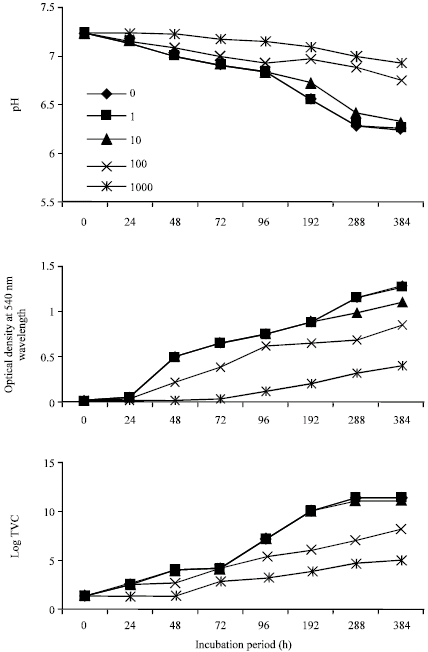 | |
| Fig. 2: | Influence of cadmium on the growth profile of Pseudomonas sp. on crude oil |
In Fig. 3, the growth rate at the exponential phase in the control culture at 96 h was 0.16 h-1 with corresponding 15.55 and 6.17 h for number of generations and generation time, respectively. In cultures containing 1000 mg L-1 cadmium exponential phase growth rate was observed at 192 h to be 0.026 h-1 with corresponding 4.98 and 38.53 h for number of generations and generation time, respectively. Exponential phase growth rate of Pseudomonas sp. in the presence of 1000 mg L-1 cadmium was observed at 72 h to be 0.7 h-1; with corresponding 5.35 and 13.46 h for numbers of generations and generation time, respectively (Fig. 4). However, the number of generations, growth rate and generation times at exponential phase in control cultures (without cadmium) were, respectively 19.63, 0.20 h-1 and 4.89 h. Number of generations increased up to 33.49 in the control as against 12.56 in cultures containing 1000 mg L-1 cadmium.
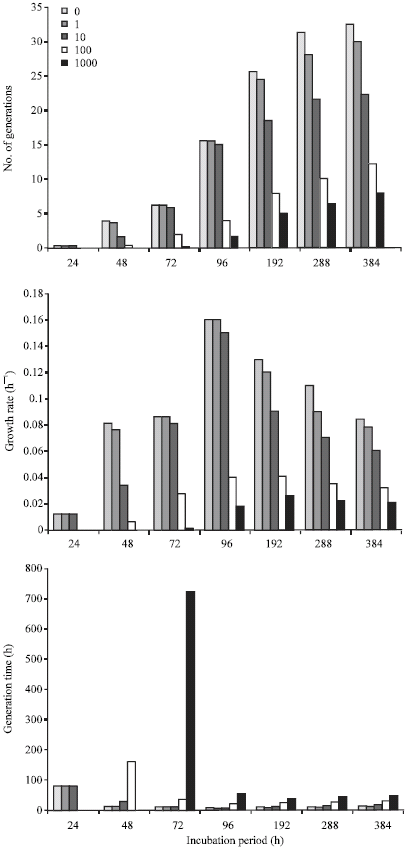 | |
| Fig. 3: | Influence of cadmium on the number of generations, growth rate and generation time of Bacillus sp. during growth on crude oil |
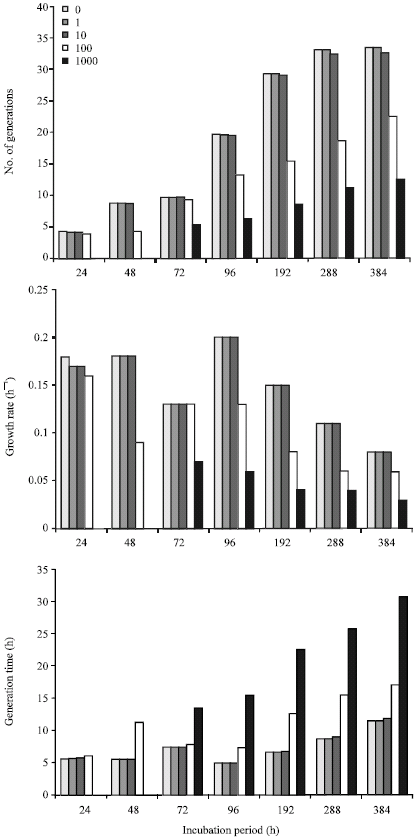 | |
| Fig. 4: | Influence of cadmium on the number of generations, growth rate and generation time of Pseudomonas sp. during growth on crude oil |
Two bacteria, Bacillus sp. and Pseudomonas sp. were used in this study. The bacteria differed firstly in their cell wall properties and secondly in the habitats from where they were isolated. While Bacillus (Gram +ve) was isolated from the benthic sediment sample with reduced amounts of oxygen, Pseudomonas (Gram -ve) was isolated from the surface waters with comparatively higher amounts of oxygen.
Growth of these organisms in liquid media containing crude oil as carbon and energy source with or without the presence of contaminating levels of cadmium resulted in a few distinctive observations.
Firstly, growth of the bacteria on crude oil resulted in a drop in the pH of growth media, as the bacterial cells metabolized the crude oil. Such changes in pH could be attributed to the production of acidic metabolites. Aerobic biodegradation of aliphatic and aromatic hydrocarbons leads to production of organic acids (Cerniglia, 1992) and goes to suggest that aerobiosis is salutary to crude oil biodegradation by these two bacterial species. However, while the pH dropped from 7.20 to 5.91 in cultures containing Bacillus sp. the observed pH drop in cultures containing Pseudomonas sp. was 7.23 to 6.23. The Bacillus sp. was observed to attack glucose fermentatively while Pseudomonas sp. attacked the sugar oxidatively during the O-F tests used for their identification. Fermentative metabolism produces more acidic end products than oxidative metabolism (Hugh and Leifson, 1953). Therefore, the high level of pH decrease observed in cultures containing Bacillus sp. could be attributed to the physiological state of the organism which was mostly fermentative.
The toxicity of cadmium to these organisms as observed over a 384 h period revealed that time was an essential factor in the mediation of this effect. Taking the organisms one at a time, it was observed that the acclimation period (defined here as the time taken to observe an initial increase in TVC of the organism) of Bacillus sp. in crude oil/MSM increased from ≤24 h to between 24 and 48 h in cultures containing 100 mg L-1 cadmium and between 48 and 72 h in those containing 1000 mg L-1. Heavy metals have been reported to lengthen acclimation periods of microorganisms (Roane and Pepper, 1997), but the duration of lengthening is dependent on metal concentration and the nature of the organism. The effect of 1 mg L-1 took 48 h to occur. This suggests that growth of this species of Bacillus, when exposed to cadmium concentrations ≤10 mg L-1 for 48 h would not be different from growth in systems without cadmium. Only prolonged exposure of the organism to metal influence would necessitate metal (cadmium) toxicity. The metal cadmium is not accumulated by the fast, unspecific, constitutive uptake systems; therefore some amount of time is needed for inducible uptake systems to be expressed. It would appear that this time reduced as cadmium concentrations increased. The growth rate of Bacillus sp. increased from 0.012 to 0.16 h-1 at 96 h in the control. Prolonged incubation resulted in decreased growth rates from 0.16 h-1 (96 h) to 0.084 h-1 (384 h). This reduction in growth rates after 96 h could be attributed to a number of factors which may include depletion of nutrients especially oxygen, build-up of recalcitrant hydrocarbon fractions and a concomitant disappearance of readily utilizable fractions and finally build-up of toxic biodegradation intermediates. Similar observations were made in cultures containing cadmium concentrations of 1, 10 and 100 mg L-1 except that the growth rates reduced as cadmium concentrations increased. In cultures containing 1000 mg L-1 cadmium, growth rate was extremely low (0.001 h-1) at 48 h. None was observed at 24 h. The generation time of the organism at 48 h was very high (722.4 h). This shows that 1000 mg L-1 cadmium concentration was an extremely toxic concentration to Bacillus sp. and rapidly selected a few resistant strains of the organism, which were able to maintain a growth rate of 0.02 h-1 from 96 h to the end of the sampling period (384 h). Although the growth rate was constant from 96-384 h, the generation times were not. This means that although the number of generations was increasing, they were not doubling with time.
In control cultures containing Pseudomonas sp. the growth rate was much higher than was observed for Bacillus sp., being 0.18 h-1 at 24 and 48 h. The drop from 0.18 to 0.13 h-1 indicates a probable disappearance of the preferred hydrocarbon fraction, which must have been used to accumulate biomass in preparation for metabolic attack on a secondarily preferred hydrocarbon substrate. Crude oil contains a number of hydrocarbon fractions, which not only differ in their chemical complexities, but also in their availabilities for or susceptibilities to microbial attack (Atlas, 1981). The toxicity of 1 mg L-1 cadmium to Pseudomonas sp. was not observed till the end of the sampling period. Cadmium concentration of 1000 mg L-1 did not produce resistant strains of Pseudomonas as was observed for Bacillus sp. The differences in toxicity might be very closely linked to cell wall differences between the two organisms as well as genetic abilities to survive in the presence of cadmium stress.
A pertinent point noted from this study was that while optical density readings in control and cultures containing 1 and 10 mg L-1 cadmium were similar up to 96 h of sampling, the total viable counts data decreased with increasing concentrations of cadmium. Count reductions were higher in cultures containing 10 mg L-1 cadmium than in 1 mg L-1 cultures. The reductions, though small, were persistent at least up to 96 h of incubation and increased with prolonged exposure to the heavy metal. Bacterial cells growing in batch culture are at different physiological stages and phases of growth at any given time and cells growing exponentially are most susceptible to the effects of a metabolic stress (Prescott et al., 1999). Most of these cells oftentimes die out while some adjust by development of resistance mechanisms to survive the stress (e.g., metal toxicity). This observation suggests that determinations of optical density alone without viable counts data in toxicity studies might not give accurate measures of microbial responses to the toxin.
Heavy metals impact the physiology of biodegrading microorganisms. The extent of such impact depends on metal concentrations, the test organism and duration of exposure to the toxic metal. Total viable counts data are more reliable than optical density measurements during toxicity tests such as those involving heavy metals.