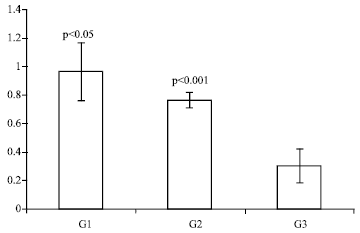
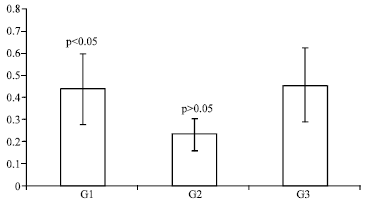
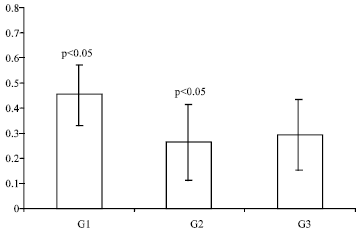
Research Article
Schistosomicidal Effect of Curcumin
Department of Medicinal Chemistry, National Research Center, Dokki, Cairo, Egypt
Mohamed M. Abdel-Aziz
Gastroenterology Center, Mansoura University
K.A. Elbakry
Damietta Faculty of Science, Mansoura University
E.A. Toson
Damietta Faculty of Science, Mansoura University
A.T. Abbas
Gastroenterology Center, Mansoura University