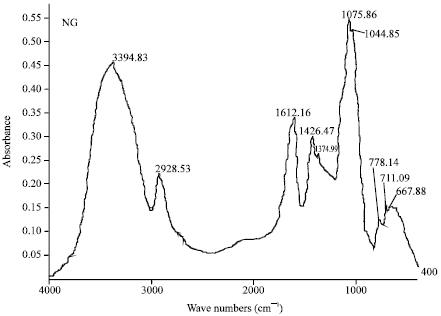
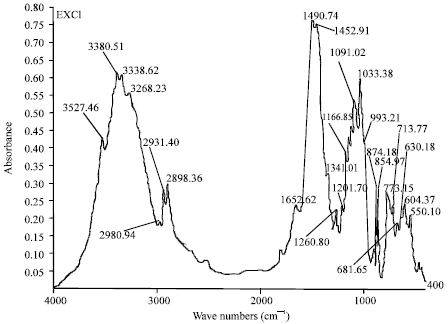
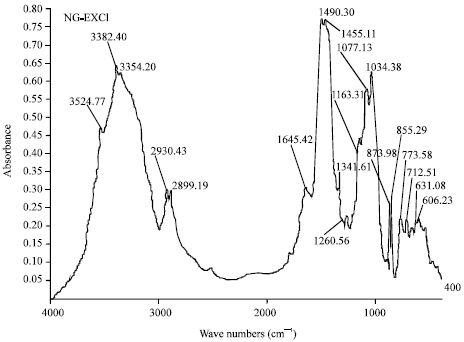
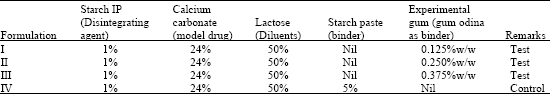Research Article
Gum Odina-A New Tablet Binder
Department of Pharmaceutical Technology, Jadavpur University, Kolkata (Calcutta) 700 032, India
Amalesh Samanta
Department of Pharmaceutical Technology, Jadavpur University, Kolkata (Calcutta) 700 032, India
Subas Chandra Dinda
Department of Pharmaceutical Technology, Jadavpur University, Kolkata (Calcutta) 700 032, India