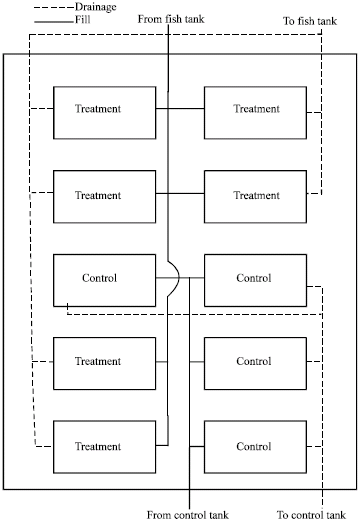
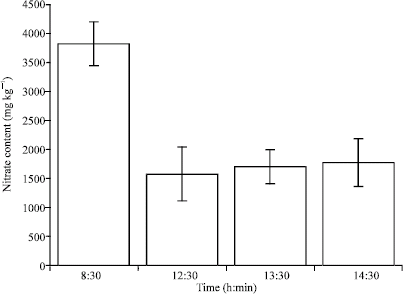
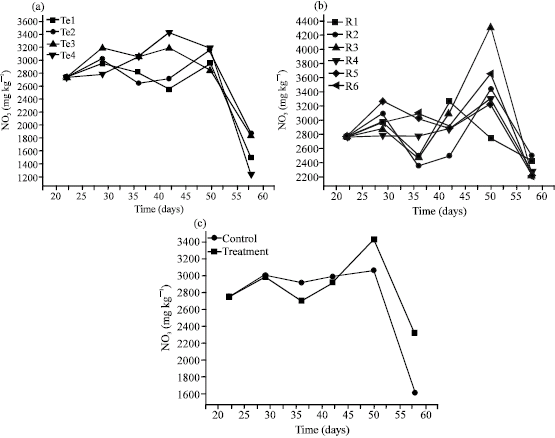
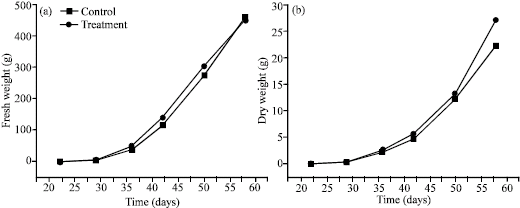
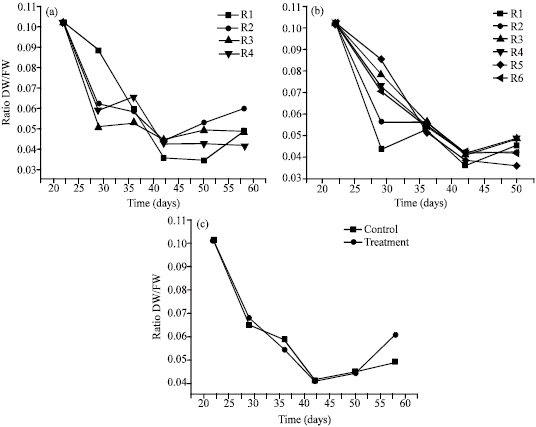
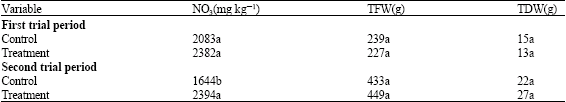
Research Article
Nitrate Content on Summer Lettuce Production Using Fish Culture Water
Division de Estudios de Posgrado, Facultad de Ingenieria, Universidad Autonoma de Queretaro, Cerro de las Campanas, Colonia las Campanas, C.P. 76010, Querataro, Qro. Mexico
V.E. Casanova-Villareal
Instituto Tecnologico de la Zona Maya, Kilometro 21.5 Carretera Chetumal-Escarcega Municipio Othon, P. Blanco, Ejido Juan Sarabia, C.P. 77060, Quintana Roo, Mexico
A. Mercado-Luna
Division de Estudios de Posgrado, Facultad de Ingenieria, Universidad Autonoma de Queretaro, Cerro de las Campanas, Colonia las Campanas, C.P. 76010, Querataro, Qro. Mexico
G.M. Soto-Zarazua
Division de Estudios de Posgrado, Facultad de Ingenieria, Universidad Autonoma de Queretaro, Cerro de las Campanas, Colonia las Campanas, C.P. 76010, Querataro, Qro. Mexico
R. G. Guevara-Gonzalez
Division de Estudios de Posgrado, Facultad de Ingenieria, Universidad Autonoma de Queretaro, Cerro de las Campanas, Colonia las Campanas, C.P. 76010, Querataro, Qro. Mexico
G. Herrera-Ruiz
Division de Estudios de Posgrado, Facultad de Ingenieria, Universidad Autonoma de Queretaro, Cerro de las Campanas, Colonia las Campanas, C.P. 76010, Querataro, Qro. Mexico
I. Torres-Pacheco
Division de Estudios de Posgrado, Facultad de Ingenieria, Universidad Autonoma de Queretaro, Cerro de las Campanas, Colonia las Campanas, C.P. 76010, Querataro, Qro. Mexico
R.V. Velazquez-Ocampo
Division de Estudios de Posgrado, Facultad de Ingenieria, Universidad Autonoma de Queretaro, Cerro de las Campanas, Colonia las Campanas, C.P. 76010, Querataro, Qro. Mexico