ABSTRACT
The presence of Atomic Oxygen (AO) in the Low Earth Orbit (LEO) environment is considered one of the main reasons for the degradation of the surfaces exposed to the Space Environment A spacecraft’s orbital velocity of 7.8 km sec-1 may expose a spacecraft to a stream of AO at energy of approximately 5 eV. At this energy, atomic oxygen initiates a number of chemical and physical reactions with the materials. The degree of surface degradation is directly proportional to AO fluence which, in turn, varies with several factors, such as: Spacecraft altitude, orientation, orbital inclination, mission duration and solar activity variation. The present paper concerns study the variation of the atomic oxygen erosion with the previous mentioned factors. In addition a comparative study of the atomic oxygen erosion effect of different spacecraft materials which are widely used for space application is performed. The atomic oxygen interaction model (ATOMOX) has been employed to evaluate the atomic oxygen flux, fluence and material erosion, based on reference atmosphere and wind models. The computation of neutral atmosphere composition is achieved using the standard NRLMSISE-00 model. Values of erosion yields have been established based on flight data. The results obtained represent that the exposed materials onboard spacecraft experience the most intense degradation at low earth orbit with lower altitudes and inclination, for surfaces in the ram direction, for prolonged exposure and at high solar activity. Also, it is found that according to the material sample understudy, Teflon shows the higher resistivity for AO erosion, for polymers; while it is Tungsten, in case of metals.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/srj.2014.1.13
URL: https://scialert.net/abstract/?doi=srj.2014.1.13
INTRODUCTION
It is known that a spacecraft in low earth orbit is exposed to an environment of a dynamic mixture of different factors that, to some degree, influence its design, such as the atomic species (in low concentrations), charged particles, vacuum, micrometeoroids, radiation, temperature extremes and man-made debris. Of particular concern in Low Earth Orbit (LEO) environment is the presence of atomic oxygen which seriously erodes many of the commonly employed spacecraft materials (Hasting and Garrett, 1996; Yokota et al., 2006; Morrison et al., 1988; White et al., 2005; Duo et al., 2011). AO erosion may degrade the thermal, mechanical and optical properties of the exposed materials. These effects can be very severe, leading to a catastrophic loss of a spacecraft. Therefore, they must be considered in the design stage (Morrison et al., 1988). In the present study we discuss the variation of the atomic oxygen erosion effect on the exposed spacecraft materials.
LEO SPACE ENVIRONMENT
Low earth environment parameters: The space environment, especially at low Earth orbit (LEO), defined for altitudes between about 200-1000 km (Rohr and van Eesbeek, 2005), is considered very hazardous for orbiting spacecraft, including Space Shuttle and International Space Station (Samwel and Hady, 2009).
The LEO space environment constituents degrade, to different degrees, the material characteristics of spacecraft being exposed to the severe LEO space environment and may harm the spacecraft enough to bring it to an unexpected end of spacecraft’s mission, caused by failure of engineering subsystem or premature reentry into the atmosphere (Allbrooks and Atkinson, 1992; Han and Kim, 2006). These constituents are:
| • | Different atomic species with Atomic Oxygen as the most abundant atom |
| • | Ionizing radiation with high energies between 5-400 MeV |
| • | Ultraviolet radiation, due to Sun's radiation which has energy flux of about 400 W/m2 |
| • | Micrometeorites of sizes from 1-50 μm in diameter and |
| • | Man-made debris of sizes from 10 μm to 1cm in diameter |
Particles concentrations in the LEO environment decrease roughly exponentially with height as illustrated in Fig. 1 which is plotted using data calculated by NRLMSIS-00 model through SPENVIS website http://www.spenvis.oma.be/. The sample profiles of the major constituent's concentrations during standard solar activity are plotted assuming nominal space (SS) orbits of circular orbit with 28.5° inclination, for one year mission length. Ordered by decreasing mass (in amu), the major constituents of the neutral environment are Argon (Ar), 39; molecular oxygen (O2), 32; molecular nitrogen (N2), 28; atomic oxygen (O), 16; Helium (He), 4; and atomic Hydrogen (H),1. Hydrostatic equilibrium makes the light particles, H and He, the dominant constituents at high altitudes, whereas heavy particles, such as Ar, O2 and N2, are relatively unimportant.
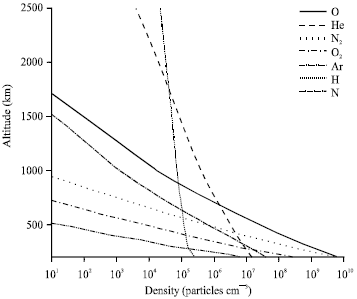 | |
| Fig. 1: | Density of the LEO environment particles as a function of altitude for one year mission length during mean solar activity |
For most practical applications, the neutral environment at LEO and polar altitudes is considered to consist mainly of Atomic oxygen. In addition, it is clarified by Fig. 1 that the atomic oxygen concentration together with the concentration of other molecules and atomic species decreases with increasing altitude.
Atomic oxygen: origin and effect: The Atomic Oxygen (AO) in the low Earth orbital environment (LEO) is formed by photo-dissociation of diatomic oxygen (Banks et al., 2004). Short wavelength (<243 nm) solar radiation has sufficient energy to break the O2 diatomic bond with energy of 12 eV in an environment where the mean free path is sufficiently long (~108 m). Thus, the probability of re-association or the formation of ozone (O3) is small which means that the rate of splitting of O2 to the atomic Oxygen Species (AO) by UV is faster than the corresponding recombination (Duo et al., 2012; Yokota et al., 2006; Rohr and van Eesbeek, 2005; White et al., 2005). As a consequence, over the LEO altitude range, atomic oxygen is the most abundant species and has a very high probability of long-term survival in the low altitudes (Banks et al., 2004; Dooling and Finckenor, 1999; Kiefer and Orwoll, 1987; Reddy, 1995). Although excited states of atomic oxygen can be formed, their lifetimes are sufficiently short. Thus, the atomic oxygen in its ground state, O(3P), dominates the LEO atomic oxygen formation, dependent upon the diatomic oxygen density and solar UV flux (Banks et al., 2004).
Oxygen atoms are known to be highly corrosive, once combining with most materials they encounter. In addition, a spacecraft’s orbital velocity of 7.8 km/sec expose spacecraft to a stream of AO at energy of approximately 5 eV (Dooling and Finckenor, 1999; Morrison et al., 1988). The AO collision with the spacecraft surfaces with this energy initiates numerous chemical and physical events on the surfaces. Erosion of surface, loss in mass, degradation in mechanical, thermal and optical properties and changes in chemical compositions of materials can be resulted through the collision with AO. In addition, materials that resist the AO erosion effects are likely to create optical glow near material surface which may hinder the visibility of the optical systems of any given space structure (Han and Kim, 2006; Shuvalov et al., 2007). Even if the flux of atomic oxygen does not erode the surface, oxidation of the surface may change the thermal properties of the surface layer (Hasting and Garrett, 1996) which would mean that the thermal requirements of the craft may be compromised by the resulting change of the equilibrium temperatures.
EROSION BY ATOMIC OXYGEN
In the present section, the erosion depth of spacecraft material is investigated depending on several factors, including Spacecraft altitude, Orientation, Orbital inclination, Mission duration and solar activity variation, taking the polyimide Kapton, common widely used material on spacecraft surfaces, as a target material. For this type of Kapton, the erosion yield; the volume of material oxidized per impacting oxygen atom, is approximately 3x10-24 cm3/atom (Hasting and Garrett, 1996) taking into consideration that the obtained results are only valid for solid material.
The erosion depth due to AO attack of exposed materials is evaluated through the calculation of the total fluence accumulated during the spacecraft mission. The atomic oxygen interaction model (ATOMOX) implement in the ESA’s Space ENVironment Information System (SPENVIS) package http://www.spenvis.oma.be/ has been employed to evaluate the atomic oxygen (and other species of the atmosphere) flux, fluence and material erosion, based on reference atmosphere and wind models. The resulting material erosion is taken as proportional to the fluence of particles on this surface.
The computation of neutral atmosphere composition is achieved using the standard NRLMSISE-00 model; it is an update of the MSIS86 model released by the U.S. Naval Research Laboratory (NRL) in 2000 and based on data of Mass Spectrometer and Incoherent Scatter radar (Hedin, 1991; Picone et al., 2002). Values of erosion yields have been established based on flight and experimental data (Banks and Rutledge, 1988).
A mean level of solar activity has been employed for the simulation, thus considering F10.7 (Previous day) = 113.7 H 10-22 Wm-2 Hz-1, F10.7 (81 days average) = 134.35x10-22 Wm-2 Hz-1 and Ap= 10 2nT. The F10.7 index is a measure of the noise level generated by the sun at a wavelength of 10.7 cm at the earth's orbit and is obtained from Solar Data at the National Geophysical Data Center (NGDC).
ftp://ftp.ngdc.noaa.gov/STP/SOLAR_DATA/SOLAR_RADIO/FLUX/Penticton_Adjusted/. The Planetary index Ap is obtained from NOAA/NWS Space Weather Prediction Center. http://www.swpc.noaa.gov/ftpmenu/indices/old_indices.html and from World Data Center for Geomagnetism, Kyoto, http://wdc.kugi.kyoto-u.ac.jp/kp/index.html.
So, in order to investigate the material degradation of spacecraft material exposed to AO flow depending on the spacecraft orbital altitude over the LEO range, Fig. 2 represents the variation of the Atomic oxygen erosion depth of Kapton as a function of spacecraft altitude, assuming orbital missions of circular orbits, having an inclination of 28.5° onto the Earth equatorial plane. The basic missions are comprised for one year mission length, between May, 16, 1998 and May, 16, 1999.
As represented in Fig. 2, the degree of surface degradation, represented here by erosion depth, is decreasing with increasing altitude which means that the exposed materials onboard spacecraft experience the most intense degradation in lower altitudes.
In LEO environment, the spacecraft orbits the Earth at velocities on the order of 7.8 km sec-1, it runs into the atomic oxygen; hence, into the ‘ram’ atomic oxygen. If the spacecraft is in an orbit that has zero inclination then the angle of attack of the atomic oxygen is perpendicular to surfaces whose surface normal points in the direction of travel. However, most spacecraft have orbits which are inclined with respect to the Earth’s equatorial plane.
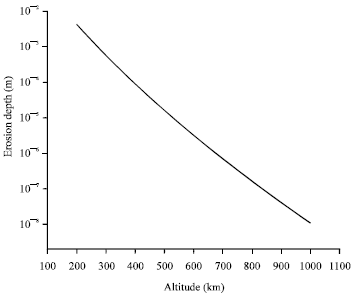 | |
| Fig. 2: | AO Erosion depth as a function of orbital altitude for mean solar activity of polyimide Kapton |
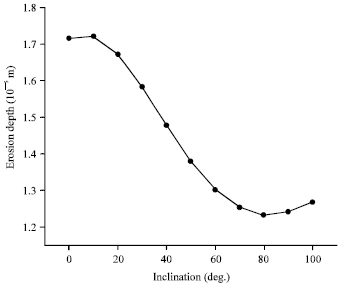 | |
| Fig. 3: | AO Erosion depth as a function of orbital inclination for mean solar activity in polyimide Kapton |
This causes the angle of attack of the arriving atomic oxygen to vary around the orbit (Banks et al., 2004). Figure 3 represents the variation of the Atomic oxygen erosion depth of Kapton as a function of spacecraft orbital inclination, assuming a circular orbit, at mid-thermospheric altitude (500 km), for mean level of solar activity. The basic missions are comprised for one year mission length, between May, 16, 1998 and May, 16, 1999.
It is shown in Fig. 3 that the degree of surface degradation, represented here by erosion depth, is decreasing with increasing inclination till reaching the polar altitudes where it begins to rising again which means that the exposed materials onboard spacecraft experience the most intense degradation in lower orbital inclination compared to polar orbits.
An additional factor that affects the spacecraft material degradation is the orientation of the exposed surface with respect to the attack of the atomic oxygen flow. Figure 4 shows the AO erosion depth as function of the surface orientation, taking the polyimide Kapton, as a target material. A circular LEO orbit with 500 km altitude and 28.5° inclination is assumed with mean level of solar activity. The orientation of the exposed surface is defined in the reference frame attached to the satellite and is defined in terms of the attitude vector which is taken in this case to be parallel to the velocity vector. As it is represented in Fig. 4, the erosion depth is greatest at the ram surface and is decreasing by increasing the orientation angle. So, surfaces in the ram direction are exposed to the most intense erosion hazard and decrease with increasing angle of incidence on surfaces away from the ram direction.
Moreover, the behavior of the surface degradation of the spacecraft materials is varying due to atomic oxygen for different mission lengths as showed in Fig. 5, assuming a circular orbit, at an altitude of 500 km and orbital inclination of 28.5°, for mean level of solar activity. The basic mission starts at May, 16, 1998. As indicated by Fig. 5, the degree of surface degradation, represented here by erosion depth, is increasing with increasing mission length. Prolonged exposure of sensitive spacecraft materials to the atomic oxygen effect may result in degraded system performance which can significantly affect mission performance and may even result in premature mission failure.
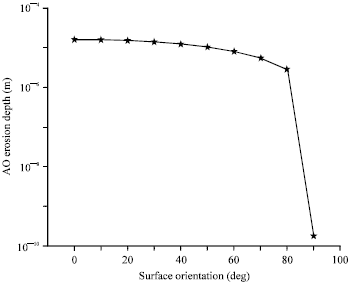 | |
| Fig. 4: | AO Erosion depth as a function of spacecraft surface orientation for mean solar activity in polyimide Kapton |
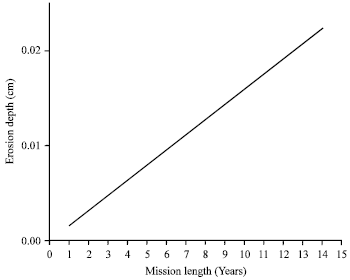 | |
| Fig. 5: | AO Erosion depth as a function of spacecraft mission length for mean solar activity in polyimide Kapton |
The atomic oxygen is formed by photodissociation and its concentration depends strongly on solar activity as the solar-caused variations in the ultraviolet radiation impinging upon the LEO atmosphere can greatly change the atomic oxygen production rate (and, therefore, the arriving flux on spacecraft surfaces). Figure 6 represents the variation of the atomic oxygen density and the corresponding AO erosion depth as a function of altitudes for different solar activity conditions, assuming a space station in a circular orbit of 500 km altitude with inclination of 28.5 degree for one year mission length and a polyimide Kapton as target material.
As shown in Fig. 6a, the atomic oxygen number density varies from 109 to 108 atoms cm-3 over an altitude range of 300-500 km during conditions of high solar activity. With low solar activity, concentrations over these altitudes are reduced to 108-106 atoms cm-3. Consequently, a spacecraft launched during times of minimum solar activity experiences less exposure to atomic oxygen than one launched during times of high activity (Fig. 6b).
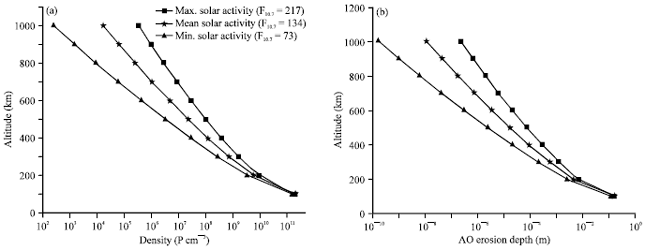 | |
| Fig. 6(a-b): | (a) AO density and (b) corresponding AO Erosion depth as function of the altitude for different solar activity levels |
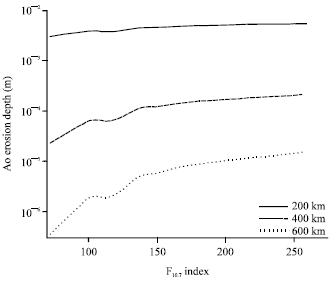 | |
| Fig. 7: | AO Erosion depth as a function of the solar activity variation represented by the F10.7 index for circular orbit, for different altitudes, with inclination of 28.5° for polyimide Kapton as target material |
Also, it is found that for higher altitudes, the solar activity has a higher influence on the erosion depth. This is represented in Fig. 6b and more clarified in Fig. 7 where the variation of the AO Erosion depth with the solar activity variation, represented by the F10.7 index is shown. The erosion depth is calculated for three different altitudes; 200, 400 and 600 km, within the LEO altitude ranges for an inclination of 28.5°, taking the polyimide Kapton as the target material. The 10.7 cm solar radio flux (F10.7) is an indicator of overall solar activity levels as a proxy for solar extreme ultraviolet radiation. At an altitude of 600 km, the difference in the erosion depth between the minimum and maximum solar activity reach about one order of magnitude which is much less in the case of lower altitude of 200 km.
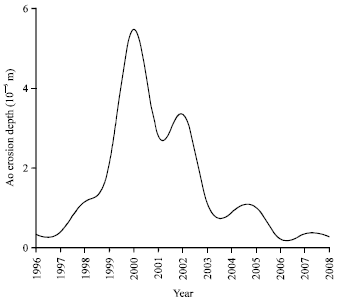 | |
| Fig. 8: | AO erosion depth variation along the solar cycle 23 for circular orbit, with altitude of 500 km, inclination of 28.5° for polyimide Kapton as target material |
| Table 1: | Low earth orbital atomic oxygen effects on spacecraft materials |
 | |
This can be explained as the solar activity has a significant effect on atmospheric density and consequently on AO density. High solar activity results in high density. Below 200 km, the density is not strongly affected by solar activity which is not the case at higher altitude where the density is greatly affected by the solar activity variation.
In addition, the average atomic oxygen fluence per year and consequently the corresponding AO erosion depth varies as a result of the solar activity consistent with the 11-year sunspot cycle. This is represented in Fig. 8 where the AO erosion depth variation along the solar cycle 23 (1996-2008) for a circular orbit with altitude of 500 km and inclination of 28.5°, taking the polyimide Kapton as the target material. The erosion depth of AO varies with varying solar activity along the solar cycle and reaches its maximum at the maximum of the solar activity which took place during the year 2000. This implies that as the Sun emits more UV and X-rays during solar maximum, it expands the upper atmosphere, thus increasing AO populations.
Finally, Table 1 summarizes the variation of atomic oxygen erosion effect on spacecraft materials at LEO environment depending on various factors.
COMPARATIVE STUDY OF THE ATOMIC OXYGEN EROSION EFFECT FOR DIFFERENT SPACECRAFT MATERIALS
Two types of materials used for satellite structures are polymers and metals. Polymers suffer more than metals when exposed to AO. Yet their outstanding properties make them popular in space applications. Polymers are preferable in the sense that they save weight due to their low densities and have better thermal insulation performance. Moreover, for electronics and optical equipment, polymers are widely used. On the other hand, metallic materials are also prone to degradation, albeit less than polymers. Metals, certainly, could maintain the optimum structural integrity (Avcu and Celik, 2003). In the present section, a comparative study of the atomic oxygen erosion effect on different polymers and metals which are widely used for space application in LEO environment is performed.
The erosion depth due to AO attack for different kinds of polymers and metals is evaluated through the calculation of the total fluence accumulated during the spacecraft mission along 10 years mission length. The ATOMOX routine of the SPENVIS package has been employed to evaluate the total daily fluence of neutral Atomic Oxygen (AO).
Quantification and susceptibility of material to atomic oxygen attack is described as an erosion yield which is the ratio of the volume or mass of material loss per each incident oxygen atom. Values of erosion yields have been established based on flight and experimental data. Banks and Rutledge (1988) present a compilation table of erosion yields of materials tested in low Earth orbit by Means of an Oxygen Ion Beam which is used by ATOMOX routine implemented in SPENVIS for both polymers and metals. The reactivity of the oxygen ion beam is higher than that observed in space. This may be partially due to higher energy which contributes to the reactivity as the energy of the oxygen ions is an order of magnitude higher than the energy of atomic oxygen in low-earth orbit.
Banks et al. (2009) in their study, developed a predictive tool to estimate the LEO atomic oxygen erosion yields of polymers. The tool has a correlation coefficient of 0.895 and an uncertainty of 6.3x10-24 cm3/atom, when compared with actual MISSE 2 PEACE polymers space data. Consequently, this predictive tool allows us, for future studies, to estimate the LEO atomic oxygen erosion yields for new polymers without requiring expensive and time consumptive in-space testing.
Table 2 represents different kinds of polymers and metals which are widely used for space purposes, in addition to their corresponding AO reactivity (erosion yield) and the used reference of each AO reactivity value. Of the most common widely used material on spacecraft surfaces, is the polyimide Kapton with erosion yield of 3x10-24 cm3/atom and titanium and tungsten metals with erosion yield of 0.0039x10-24 cm3/atom and of 0.00044x10-24 cm3/atom, respectively.
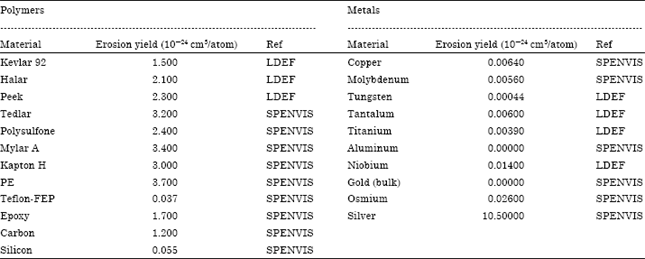
Figure 9 draws the AO erosion depth as a function of the AO fluence for different polymers as target materials onboard spacecraft in a LEO environment. For more clarification, a separate graph for silicon and Teflon FEP as target materials is included in Fig. 9. A circular orbit of 500 km altitude and inclination of 28.5° is assumed. It is found that both Teflon FEP and silicon show highest resistivity for AO erosion effect in comparable to the other polymeric materials understudy with a merit for Teflon than silicon, taking into consideration that the reactivity of FEP differs for different sources as it ranges from 0.05 to 10x10-24 cm3/atom which would vary its resistitive to AO erosion. Also, it is clear that Si is safe as AO combines to generate SiO2 (the shielding glass). Conversely, Polyethylene shows the lower resistivity for AO erosion effect.
| Table 2: | Target materials and their corresponding ao reactivity for both polymers and metals |
 | |
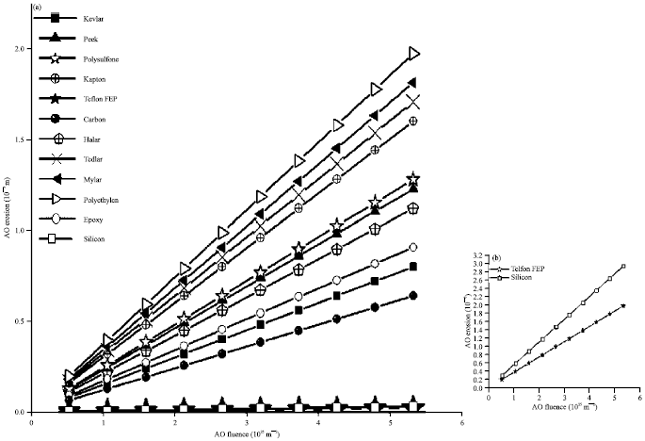 | |
| Fig. 9(a-b): | AO Erosion depth as a function of the AO fluence for different polymers as target materials onboard a spacecraft |
On the other hand, Fig. 10 draws the AO erosion depth as a function of the AO fluence for different metals as target materials onboard spacecraft in a LEO environment with separate graph for Silver as a target material. A circular orbit of 500 km altitude and inclination of 28.5o is assumed.
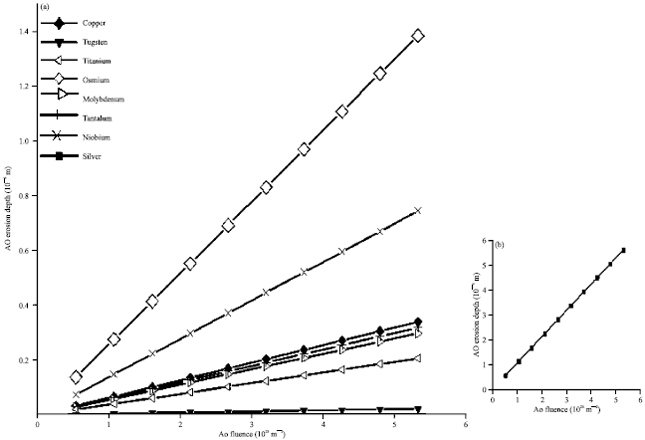 | |
| Fig. 10(a-b): | AO Erosion depth as a function of the AO fluence for different metals as target materials onboard a spacecraft |
As it is represented by Fig. 10, the AO erosion depth of Tungsten is smallest in comparable with the other metals understudy while Silver shows the lowest resistance for the AO erosion effect.
The reaction of atomic oxygen with silver may be quite high which is considered an exception. Although the usefulness of silver in solar-array and optical components applications because of their high conductivity and reflectivity, Silver possesses relative high capture efficiency for AO. On exposure to AO flux, the top of silver surface layer is oxidized, causing loss of both electrical conductivity and mechanical strength and after prolonged exposure, the oxide layer are not adhere off to but spall off the silver substrate (Zhang et al., 2002; Duo et al., 2012). So, protective coating to Silver is used in order to provide a barrier between the base material and the AO environment.
Moreover, Fig. 9 and 10 show that in general, Polymers suffer more than metals when exposed to Atomic Oxygen erosion effect, based on the employed models.
CONCLUSION
The present article, studies the variation of atomic oxygen erosion effect with various parameters, including spacecraft altitude, orientation, orbital inclination, mission duration and solar activity variation. The ATOmic OXygen interaction model (ATOMOX) implement in the ESA’s Space ENVironment Information System (SPENVIS) package http://www.spenvis.oma.be/ has been employed to evaluate the atomic oxygen flux, fluence and material erosion and the standard NRLMSISE-00 model is used to compute the neutral atmosphere composition. The results obtained are summarized as follows:
| • | The exposed materials onboard spacecraft experience the most intense degradation in lower altitudes |
| • | The exposed materials onboard spacecraft experience the most intense degradation onboard spacecraft in orbits with small inclination compared to polar orbits |
| • | Surfaces in the ram direction are exposed to the most intense erosion hazard, with exposure decreasing with increasing angle of incidence on surfaces away from the ram direction |
| • | Prolonged exposure of sensitive spacecraft materials to the atomic oxygen effect may result in degraded system performance which can significantly affect mission performance and may even result in premature mission failure |
| • | A spacecraft launched during times of minimum solar activity experiences less exposure to atomic oxygen than one launched during times of high activity |
| • | The Solar activity has a higher influence on the erosion depth for high altitudes in comparable with lower altitudes |
| • | The average atomic oxygen fluence per year and consequently the corresponding AO erosion depth varies as a result of the solar activity consistent with the 11-year sunspot cycle |
Thus, spacecraft should be provided with AO protection optimized for the AO environment in which they will operate for the planned mission life.
Furthermore, a comparative study of the atomic oxygen erosion effect of different spacecraft materials including polymers and metals in LEO environment is performed. It is found that both Teflon FEP and silicon show higher resistivity for AO erosion effect in comparable to the other polymeric materials understudy, using the low SPENVIS FEP reactivity. Conversely, Polyethylene shows the lower resistivity for AO erosion effect. Moreover, it appears that the AO erosion depth of Tungsten is smallest in comparable with the other metals understudy while Silver shows the lowest resistance for the AO erosion effect. Finally, it appears that Polymers suffer more than metals when exposed to AO, based on the models employed, implemented in SPENVIS.
REFERENCES
- Duo, S.W., M.M. Song, T.Z. Liu and M.S. Li, 2012. SiO2 coatings prepared by sol-gel process protecting silver from atomic oxygen erosion. Applied Mech. Mater., 121-126: 3044-3047.
CrossRef - Han, J.H. and C.G. Kim, 2006. Low earth orbit space environment simulation and its effects on graphite/epoxy composites. Compos. Struct., 72: 218-226.
CrossRef - Hedin, A.E., 1991. Extension of the MSIS Thermosphere Model into the middle and lower atmosphere. J. Geophys. Res.: Space Phys., 96: 1159-1172.
CrossRefDirect Link - Picone, J.M., A.E. Hedin, D.P. Drob and A.C. Aikin, 2002. NRLMSISE-00 empirical model of the atmosphere: Statistical comparisons and scientific issues. J. Geophys. Res., Vol. 107.
CrossRef - Reddy, M.R., 1995. Effect of low earth orbit atomic oxygen on spacecraft materials. J. Mater. Sci., 30: 281-307.
CrossRefDirect Link - Samwel, S.W. and A.A. Hady, 2009. Space radiation environment forecast for EGYPTSAT-2 satellite. Space Weather, Vol. 7.
CrossRef - Shuvalov, V.A., G.S. Kochubei, A.I. Priimak, N.I. Pismennyi and N.A. Tokmak, 2007. Changes of properties of the materials of spacecraft solar arrays under the action of atomic oxygen. Cosmic Res., 45: 294-304.
CrossRefDirect Link - Duo, S.W., M.M. Song, Y. Luo, T.Z. Liu and W.M. Gao, 2011. Atomic oxygen effects of polyimide/silica hybrid films in low earth orbit environment. Adv. Mater. Res., 177: 686-689.
CrossRefDirect Link - White, C.B., G.T. Roberts and A.R. Chambers, 2005. Measurement of 5-eV atomic oxygen using carbon based films: Preliminary results. IEEE Sensors J., 5: 1206-1213.
CrossRef - Zhang, L., C.W. Yan, Q. Qu, G. Sun, J.Y. Tong and C.N. Cao, 2002. Ground-based investigation of atomic oxygen effects on naked Ag and Ag with protective organic coatings. Trans. Nonferrous Metals Soc. China, 12: 869-873.
Direct Link








