Research Article
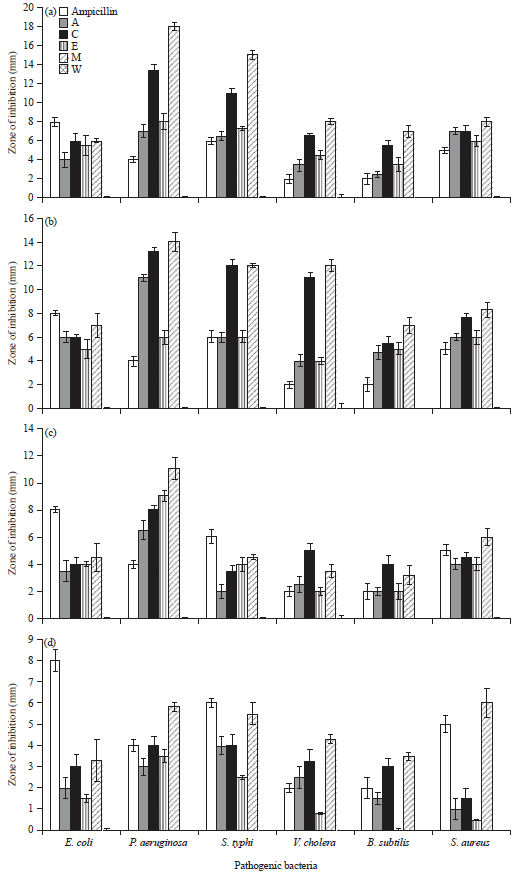
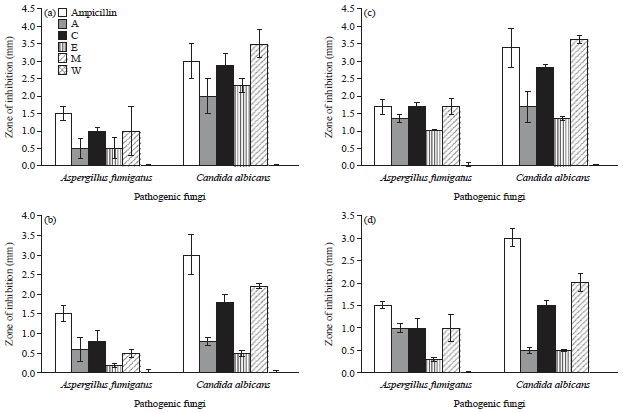
Screening the Antimicrobial Activity of Different Sepia officinalis (Cephalopoda: Sepioidea) Parts Collected from Alexandria Mediterranean Waters, Egypt Against Some Human Pathogens
Marine Environmental Division, National Institute of Oceanography and Fisheries, 21556 Alexandria, Egypt
LiveDNA: 20.18969
Rafik Riad
Marine Environmental Division, National Institute of Oceanography and Fisheries, 21556 Alexandria, Egypt