Research Article
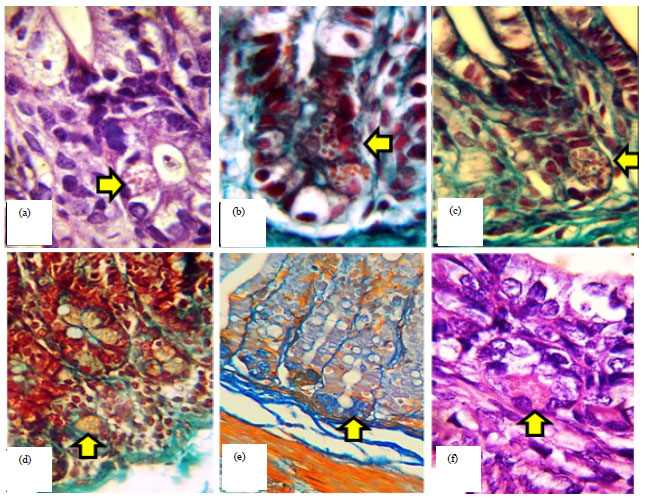
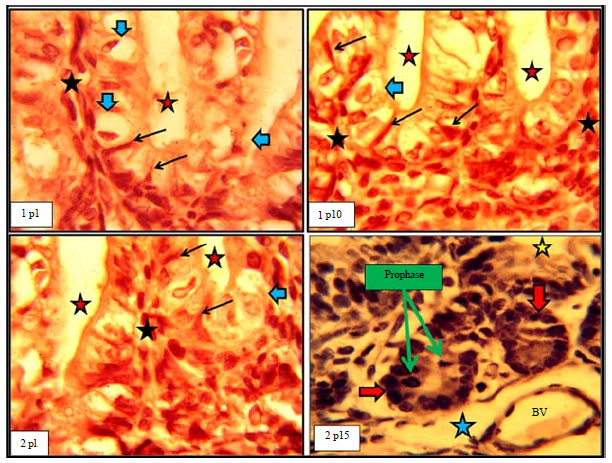
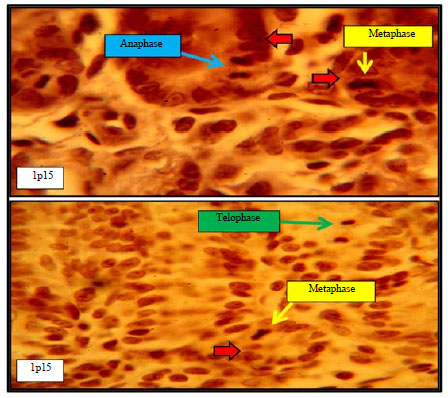
Histomorphological Relationship of Paneth Cells with Stem Cells in the Small Intestine of Indigenous Rabbit at Different Postnatal Ages
Department of Anatomy, College of Veterinary Medicine, Baghdad University, Baghdad, Iraq
LiveDNA: 964.4860
A.G. Al- Haaik
Department of Anatomy, College of Veterinary Medicine, University of Mosul, Mosul, Iraq
LiveDNA: 964.14952