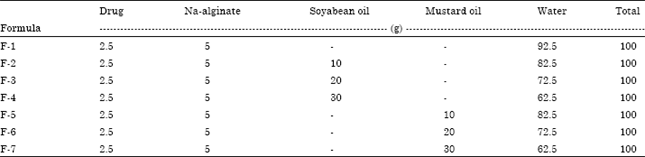
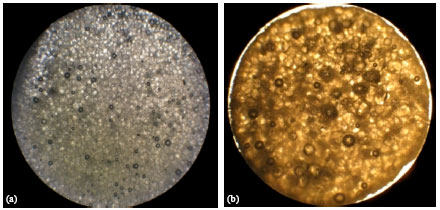
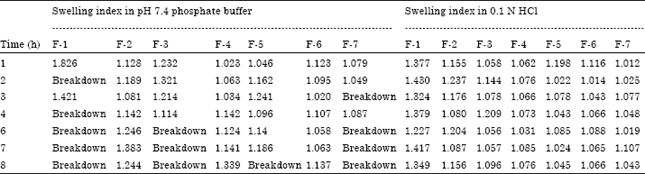
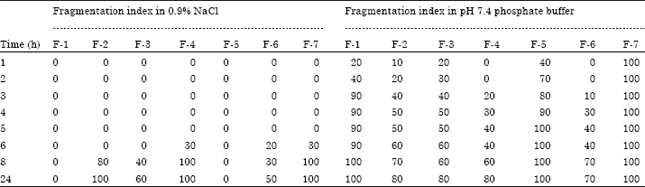
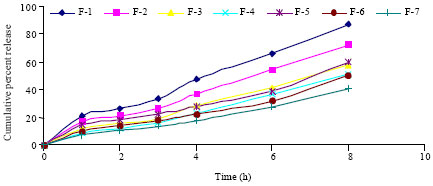
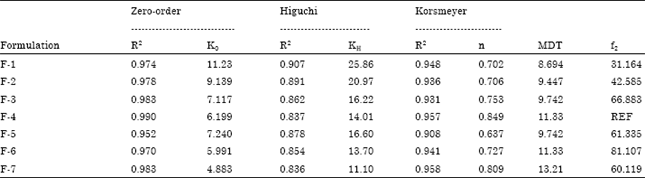
Research Article
Development and Evaluation of Ionotropically Emulsion Gelled Sodium Alginate Beads and its Morphological Characterization by Optical Micrographs
Department of Pharmacy, The University of Asia Pacific, Dhaka-1209, Bangladesh
Tasnuva Haque
Department of Pharmacy, Stamford University Bangladesh, 51 Siddeswari Road, Dhaka-1217, Bangladesh
Deepannita Barua
Department of Pharmacy, The University of Asia Pacific, Dhaka-1209, Bangladesh