Research Article
Preliminary Observations Using Canine Parvovirus-Specific Transfer Factor in the Prevention of Canine Parvovirus Disease
College of Animal Science and Technology, Yangzhou University, Yangzhou, China
M.H. Ye
College of Bioscience and Biotechnology, Yangzhou University, Yangzhou, China
R. Chen
College of Animal Science and Technology, Yangzhou University, Yangzhou, China
J.T. Ding
College of Animal Science and Technology, Yangzhou University, Yangzhou, China









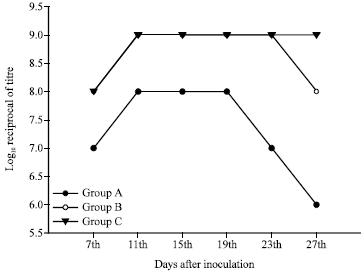
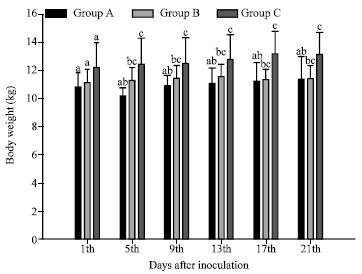
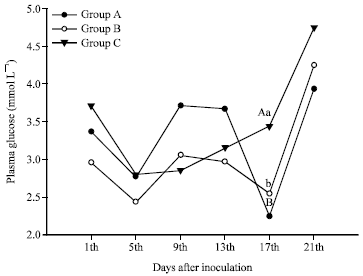
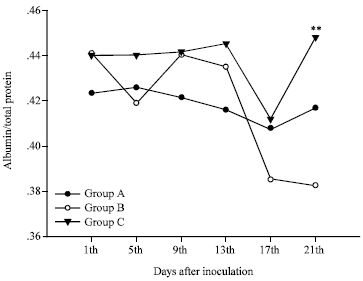
Rose Garcia Reply
Thank you for doing this study and having the results appear to be read on the internet.