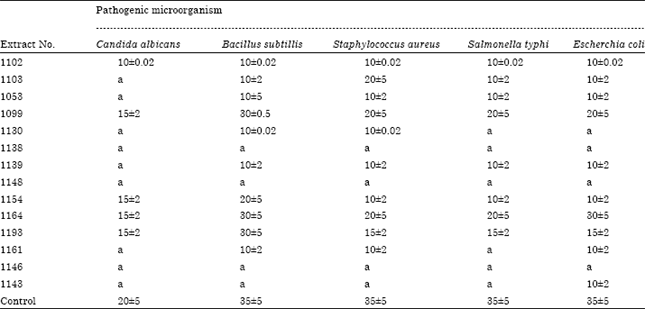
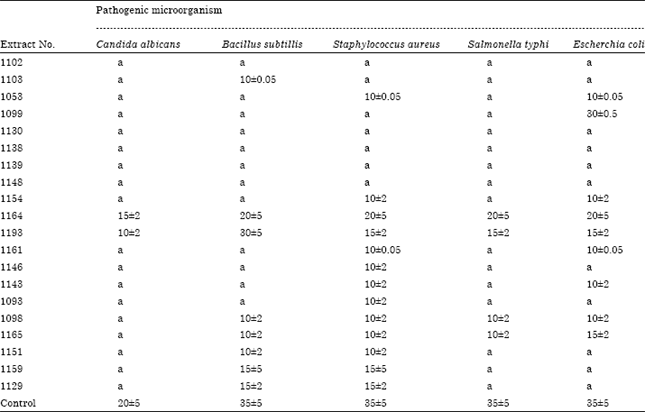
Short Communication
Antimicrobial Properties of Penicillium Species Isolated from Agricultural Soils of Northern Iran
Department of Pharmacognosy and Biotechnology, Faculty of Pharmacy, Mazandaran University of Medical Sciences, Sari, Iran
M. A. Tajick-Ghanbary
Department of Mycology and Plant Pathology, College of Agronomic Sciences, Sari Agriculture and Natural Resources University, Iran
N. Shahrokhi
Department of Pharmacognosy and Biotechnology, Faculty of Pharmacy, Mazandaran University of Medical Sciences, Sari, Iran