Research Article
Incidence of Fusarium Toxins in Rice from Karnataka, India
Department of Fermentation Technology and Bioengineering, Central Food Technological Research Institute, Mysore, Karnataka, 570 020, India
E.R. Rati
Department of Human Resources Development, Central Food Technological Research Institute, Mysore, Karnataka, 570 020, India
H.K. Manonmani
Department of Fermentation Technology and Bioengineering, Central Food Technological Research Institute, Mysore, Karnataka, 570 020, India









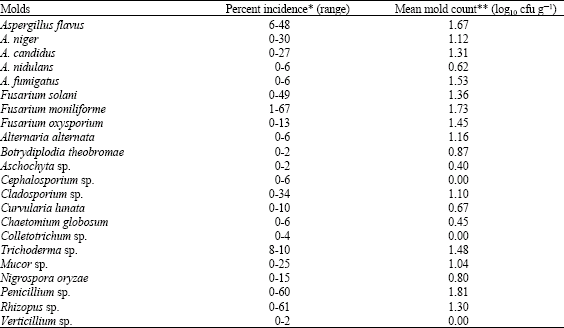
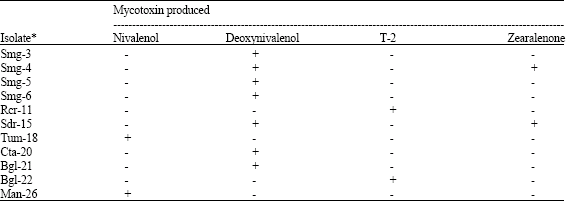

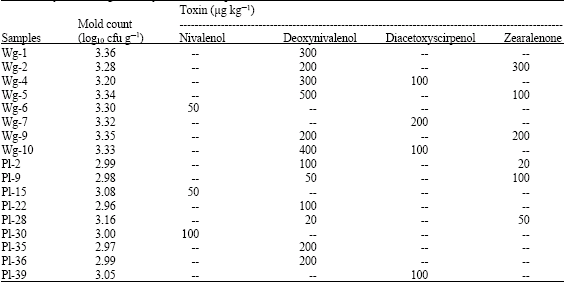

molly Knight Reply
thank you for this information. Can you tell me what the symptoms may be with ingestion of these molds?
Many thanks for your time
Molly