Research Article
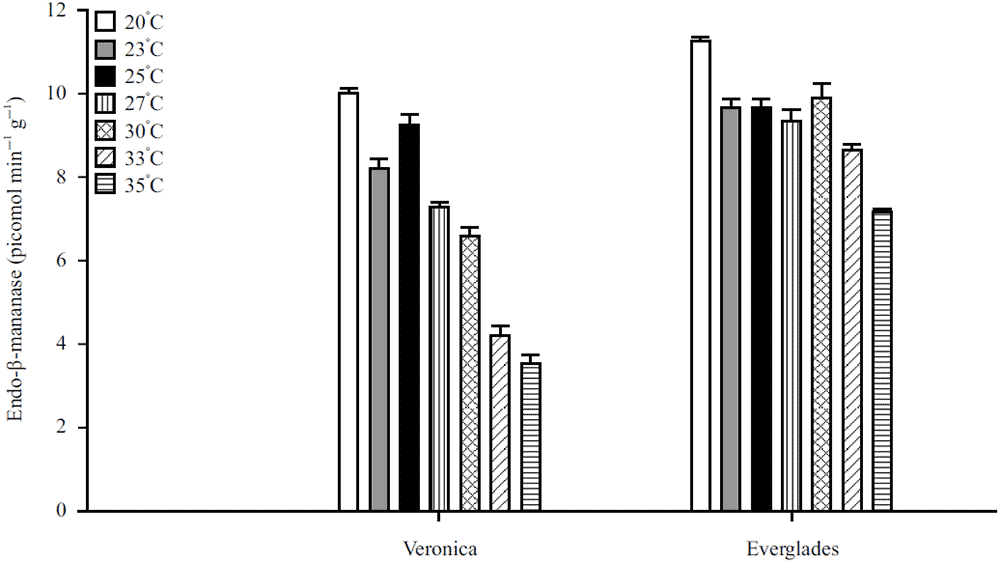
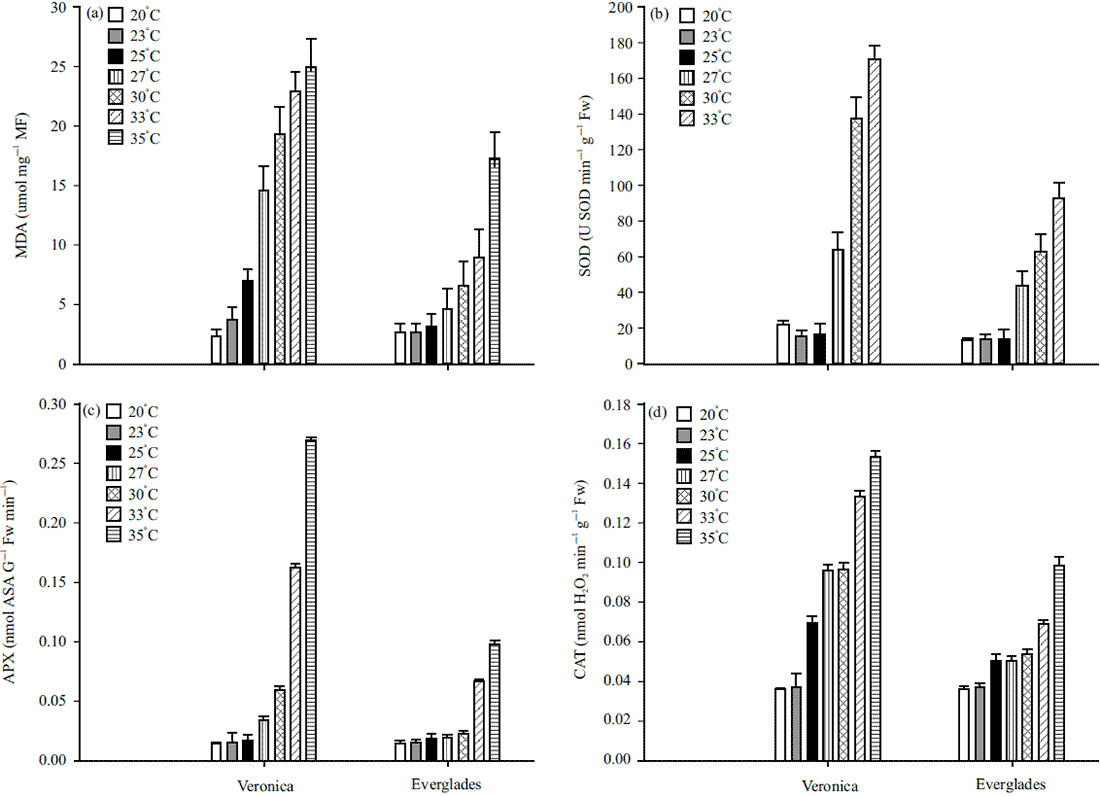
Endo-β-Mannanase Enzyme Activity is Related to Thermo Inhibition in Lactuca sativa Seeds
Federal University of Lavras, Lavras, Aquenta Sol, 37200-900, Lavras, Minas Gerais, Brazil
H.O. Santos
Federal University of Lavras, Lavras, Aquenta Sol, 37200-900, Lavras, Minas Gerais, Brazil
L.A.A. Gomes
Federal University of Lavras, Lavras, Aquenta Sol, 37200-900, Lavras, Minas Gerais, Brazil
T.B. Fantazzini
Federal University of Lavras, Lavras, Aquenta Sol, 37200-900, Lavras, Minas Gerais, Brazil
E.V.R. Von-Pinho
Federal University of Lavras, Lavras, Aquenta Sol, 37200-900, Lavras, Minas Gerais, Brazil
R.M.O. Pires
Federal University of Lavras, Lavras, Aquenta Sol, 37200-900, Lavras, Minas Gerais, Brazil
E.R. Carvalho
Federal University of Lavras, Lavras, Aquenta Sol, 37200-900, Lavras, Minas Gerais, Brazil