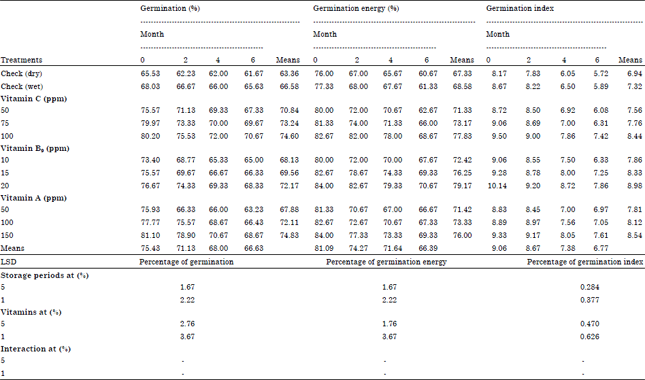
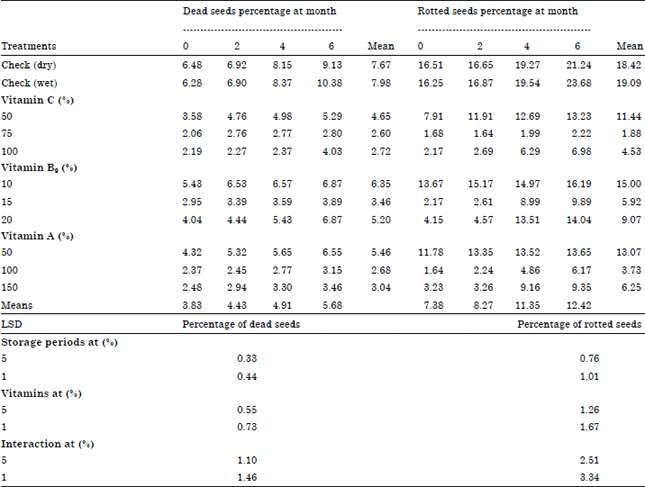
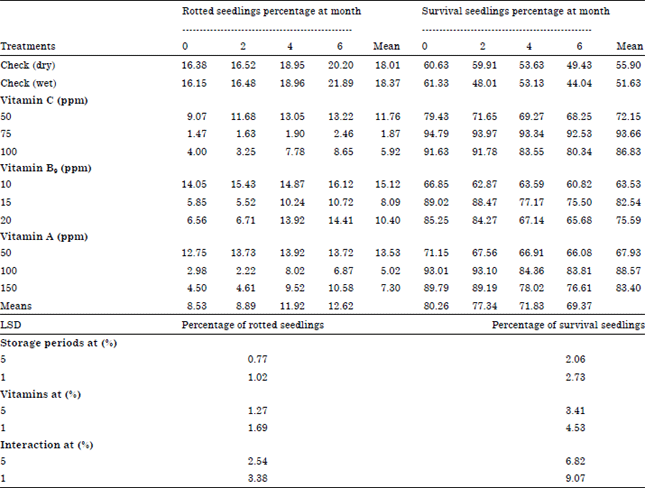
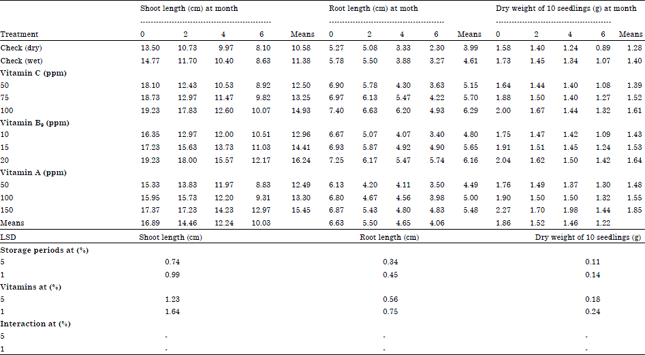
Research Article
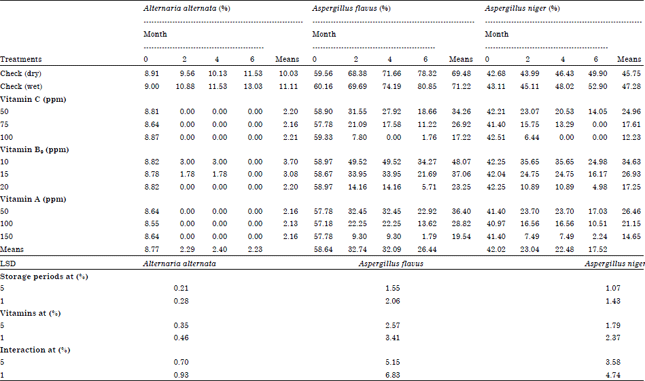
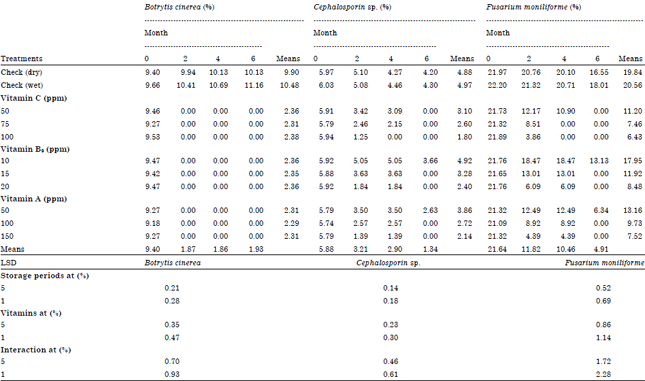
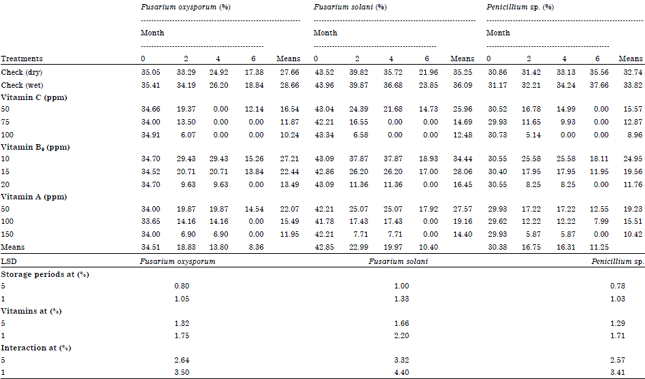
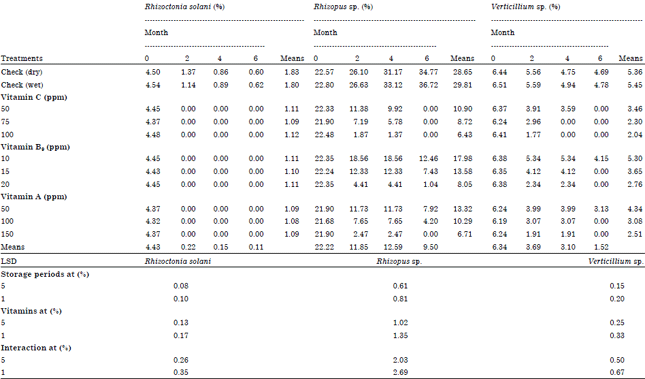
Role of Some Vitamins in Decreasing of Soybean Seed Deterioration During Storage
Department of Mycological Research, Plant Pathology Research Institute, ARC, Egypt
Aml E.A. El-Saidy
Department of Seed Technology Research, Field Crop Research Institute, ARC, Egypt
K.M. Abd El-Hai
Department of Leguminous and Forage Crop Diseases, Plant Pathology Research Institute, ARC, Egypt