Review Article
Seed Scarification Methods and their Use in Forage Legumes
Department of Plant Sciences, University of Wyoming, Laramie, Wyoming 82071, USA
M.A. Islam
Department of Plant Sciences, University of Wyoming, Laramie, Wyoming 82071, USA
Legumes are a member of the Fabaceae family which contains 700 genera and about 19,000 species (Graham and Vance, 2003). They are distinctive with unique floral structures, pod-like fruits and nodule formation systems in the root. Many species from this family have been economically important to humans as far back 3,000 year ago when wild Fabaceae family was domesticated in America and Asia (Kaplan and Lynch, 1999). By the early 21st century, legume production has occupied approximately 15% of the earth’s surface (Graham and Vance, 2003). The significant increase of legume land use is mainly due to production of high quality forage (Evers, 2011), ability to improve soil quality by fixing atmospheric N (Faria et al., 1989) and the extension of spring growth (Balasko and Nelson, 2003). A good example of an attractive forage legume is Medicago species (e.g., Medicago sativa L.), the most widely and extensively used representative forage legume in the world (Bouton, 1996). Other important forage legumes include Trifolium and Astragalus species (Carleton et al., 1971; Acharya et al., 1993; Patane and Bradford, 1993; Martin and De La Cuadra, 2004). In many instances, N fixation by Trifolium species is significant. For example, red clover (T. pretense L.) and white clover (T. repens L.) are able to fix up to 373 and 545 kg N ha-1 year-1, respectively (Carlsson and Huss-Danell, 2003). Recently, Astragalus species such as cicer milkvetch (CMV: A. cicer L.) has attracted attention of many scientists and producers. This is a perennial legume, highly tolerant to overgrazing because of its vigorous root systems (Tilley et al., 2008) and does not produce bloat problem in ruminant animals (Twidwell and Kephart, 2002; Acharya et al., 2006).
Despite the great importance and characteristics, establishment of forage legumes is difficult. One of the major constraints in successful stand establishment of forage legumes is hard seed. High hard seed content in a seed lot can cause delayed or decreased seedling emergence. As a result, stands become thin, sporadic and less competitive with weeds or undesirable species. Such legume stands reduce not only N fixation but also lower yield and quality. Therefore, reduction of hard seed content in a seed lot of forage legumes is important before planting. Seed scarification, a physical damage to break the hard seed coat without lowering the quality of seeds, has been studied for more than a century (Dixon, 1901; Harrington, 1916; Stewart, 1926; Rincker, 1954; Rolston, 1978; Stanwood, 1980; Rutar et al., 2001; Zeng et al., 2005; Dittus and Muir, 2010).
Seed scarification methods have been developed and modified over time to make these more practical and effective. Important methods of seed scarification include heat, freeze-thaw, mechanical and acid scarification. Heat scarification is the method that uses high temperatures to break or crack seed coat (Staker, 1925; Tomer and Maguire, 1989). Freeze-thaw scarification is a method that breaks the seed coat by exposing seeds to temperature alternations between low and high (Stout, 1990; Rutar et al., 2001). Mechanical scarification is a technique to physically create scars on seed surface to increase water imbibition of the seed (Uzun and Aydin, 2004; Rostami and Shasavar, 2009; Olisa et al., 2010; Jayasuriya et al., 2012). Acid scarification is a chemical method to melt seed coat and soften hard seed (Can et al., 2009). The objective of the present study is to review and summarize some important seed scarification methods and their use in forage legume species, especially in Medicago, Trifolium and Astragalus species, aiming to improve productivity and profitability of these valuable forage legumes. The information generated in the review is useful not only for researchers and producers but also for seed companies.
HEAT SCARIFICATION
Heat scarification has been one of the most popular methods because it is simple and easy to use. Two main heating devices used in heat scarification include oven (Harrington, 1916; Staker, 1925; Stewart, 1926; Lute, 1927; Rincker, 1954; Rutar et al., 2001) and hot water bath (Uzun and Aydin, 2004; Patane and Gresta, 2006; Can et al., 2009). Efficacy of heat scarification varies significantly depending on heating devices, treatment times and temperatures.
Heat scarification using the dry heat in oven seems to be effective on hard seed reduction and germination improvement when appropriate treatment time and temperature are used (Harrington, 1916; Staker, 1925; Stewart, 1926; Lute, 1927; Rincker, 1954; Tomer and Maguire, 1989; Rutar et al., 2001). Various temperatures (below 40°C to over 100°C) and treatment times (one min to 21 h) were used in different studies and variable results were reported. Some treatments were more effective than the others. For example, heat scarification had little influence on hard seed reduction when treatment temperatures were between 40 and 50°C (Harrington, 1916; Lute, 1927; Rutar et al., 2001). Neither reduction of hard seed (range 14-29%) nor improvement of germination (range 87-96%) was observed in seeds of three cultivars (‘Osjecka 10’, ‘Osjecka 88’ and ‘Slavonka’) of alfalfa (M. sativa L.) when they were heated at 40°C for 4 h (Rutar et al., 2001). Heat scarification had no influence on seeds of red clover and sweet clover (Melilotus officinalis L. Pall.) when they were subjected to treatment temperature at 50°C for 21 h (Harrington, 1916). Lute (1927) also reported that temperature below 50°C had no influence on alfalfa seeds.
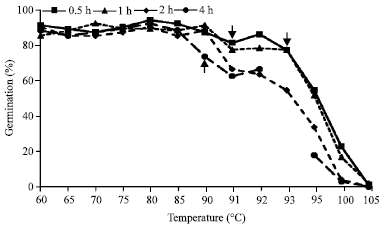 | |
| Fig. 1: | Effect of heat scarification on alfalfa seeds with various temperatures and four treatment timings. Arrows indicate the death point of alfalfa seeds in each treatment time. There is a missing value for the 4 h treatment at 93°C (Staker, 1925) |
Higher temperatures may be more effective on hard seed reduction compared to the lower temperatures; however, negative impact was observed when temperatures were higher than 80°C (Rincker, 1954; Rutar et al., 2001). Greater amount of dead seeds (38%) were observed when alfalfa seeds were heated at 80°C for one h, while lesser dead seeds (1%) were found at 40°C when heated for 5 h (Rutar et al., 2001). Alfalfa seeds were completely damaged when heated at 104°C for 1 h (Rincker, 1954). Staker (1925) conducted a study to investigate mortality of alfalfa seeds during heat scarification. In this study, alfalfa seeds were heated over four different times (half, one, two and four h) at various temperature regimes starting from 60 to 105°C. Germination declined at temperatures greater than 80°C and the number of dead seeds increased when temperatures were 90, 91 and 93°C with treatment times of four, one and two and half h, respectively (Fig. 1).
Nevertheless, heat scarification with high temperatures was shown to be effective and successful in improving germination in many studies. For example, Rincker (1954) scarified alfalfa seeds using heat scarification at 104°C with treatment time of one, two, three and four min. Germination was improved greatly from 47% in untreated control to 84% after four min of heat scarification. Stewart (1926) showed that heat scarification at 80°C was effective when heated for 2 h in dry heat as it increased germination of alfalfa seeds from 69 in untreated control to 99%. He also reported that weed seed could be filtered from alfalfa seed lots by applying heat scarification. Weed seeds contaminated in alfalfa seeds were killed when heated at 85°C for 4 h while germination of alfalfa seeds was increased. Such weed species were dodder (Cuscuta sp.), atriplex tumbleweed (Atriplex hastate L.) and Russian thistle (Salsoli tragus L.). Varying effectiveness of heat scarification is reported when treatment temperatures are higher than 80°C. As compared to the higher temperatures, there were no observations of abnormal seedlings or dead seeds when heat scarification was conducted with temperatures higher than 60°C and below 80°C (Staker, 1925; Tomer and Maguire, 1989). Hard seed of alfalfa was successfully reduced from 21% in control to 3% when heated at 60°C for 2 h in an oven, on the other hand, germination was improved from 72 in untreated control to 91% (Staker, 1925; Tomer and Maguire, 1989). The effectiveness of heat scarification with the use of oven was tested recently for winter annual medic (M. rigidula) and CMV (Islam and Kimura, 2010). The seeds of ‘Laramie’ medic and three cultivars of CMV (‘Monarch’, ‘Oxley’ and ‘Lutana’) were heated at 60°C in an oven. Treatment timings used were one, two, three, four and five h. The heat scarification neither reduced hard seed nor improved germination of medic and two cultivars (Oxley and Lutana) of CMV.
 | |
| Fig. 2(a-b): | Comparison of palisade layer of (a) M. orbicularis (511 x) and (b) A. hamosus (596 x); adapted from Patane and Gresta (2006). M. orbicularis has thicker seed coat than A. hamosus |
However, hard seed reduction (61% in untreated control to 11% after 1 h in the heat scarification) and germination improvement (28% in untreated control to 76% after 1 h in the heat scarification) was observed in Monarch CMV. The observations by Tomer and Maguire (1989) and Islam and Kimura (2010) suggested that the effectiveness of heat scarification varies depending on genus and species and even species with the same genus (M. sativa vs. M. rigidula) and cultivars within the same species (Oxley and Lutana vs. Monarch). This variation may be caused by the difference in seed coat structures. For example, Patane and Gresta (2006) reported that the palisade layer, responsible for water resistance of seeds, of M. orbicularis seeds was approximately twice as thick as that of A. hamosus seeds (Fig. 2); as a result, seeds of M. orbicularis were less affected by hot water and sulfuric acid scarification than A. hamosus (milkvetch) seeds. Oxley and Lutana may have similar seed coat structures. In other words, these two cultivars may have similar thickness and composition of seed coat providing similar hard seed contents. These structural differences may be due to the genetics of the species and cultivars or environment in which the seeds were produced or a combination of both.
Unlike the heat scarification using oven, heat scarification with a hot water bath has not been shown to be effective on hard seed reduction of many Medicago species. Patane and Gresta (2006) soaked button medic (M. orbicularis L. Bartal) and milkvetch seeds in a hot water bath with five different temperatures, starting from 60 to 100°C at an interval of 10°C. These treatments had no effect on button medic seeds but had great effect on milkvetch. Hard seed of milkvetch was reduced from 93 in untreated control to 8% when treated at 80°C for 10 min. Can et al. (2009) also reported that hot water bath treatment had no influence on hard seed of button medic even when seeds were heated at 90°C. It has also been reported that hot water bath treatment had little effect on other Medicago species such as M. hispida, M. arabica, M. falcata (Uzun and Aydin, 2004), M. rotate, M. turbinate, M. scutellata (Can et al., 2009) and M. marina L. (Scippa et al., 2011). In contrast, germination of M. arborea was increased from 45 in untreated control to 71% when the seeds were soaked in a hot water bath at 100°C for four min (Travlos and Economou, 2006). The positive effect of hot water bath treatment was seen in the legume species common in arid environment (Travlos and Economou, 2006; Travlos et al., 2007).
FREEZE-THAW SCARIFICATION
Mechanism behind the reduction of hard seededness by a freeze-thaw scarification is to make tiny scars on hard seed coat and make seed coat brittle to enhance germination (Busse, 1930; Pritchard et al., 1988; Stout, 1990; Hall et al., 1993). A force that produces scars on seed surface through this technique is depending on the size, shape and water content of seeds and treatment intensity and durations (Candolle, 1922; Steinbauer, 1926; Busse, 1930). Methods for cooling in freeze-thaw scarification include freezer (Midgley, 1926; Shibata and Hatakeyama, 1995); carbon dioxide (CO2) snow, dry ice, liquid air (Busse, 1930; ultra-low freezer (Stout, 1990); acetone (Rutar et al., 2001); liquid N (Brant et al., 1971; Stanwood, 1980; Pritchard et al., 1988; Acharya et al., 1993; Patane and Gresta, 2006).
Researchers have reported that temperatures at -80°C or lower may be more effective than temperatures greater than -80°C (Midgley, 1926; Busse, 1930; Stout, 1990). When alfalfa seeds were subjected to freeze-thaw scarification with cooling temperatures at -5°C or at -15°C for 36 h and with warming at room temperatures for six d (one cycle), germination was not improved (Midgley, 1926). In another study, milkvetch seeds were stored in freezer at -22°C for 2, 4, 7, 30, 60, 90 and 180 d, however the highest germination was below 78% after this freeze-thaw scarification (Shibata and Hatakeyama, 1995). Busse (1930) compared effectiveness of three freezing temperatures, -20°C (by dry ice), -80°C (by CO2 snow) and -190°C (by liquid air). Freeze-thaw cycle was completed when the cooled seeds were placed at room temperature (slow warming) or placed on a warm metal plate (rapid warming). Germination was increased from 53, 62 and 60% in untreated control to 86, 78 and 98% when treated with dry ice, CO2 snow and liquid air with rapid warming, respectively. Repeated freeze-thaw cycles were necessary to improve germination of alfalfa seeds when dry ice was used.
Repeating freeze-thaw cycle seems to be more effective in making a fragile seed coat (Busse, 1930; Stout, 1990; Rutar et al., 2001). For example, Stout (1990) conducted a study in two cultivars of alfalfa (‘Peace’ and ‘Anik’) with freeze-thaw scarification at -80°C for cooling (by ultra-low bio freezer) and 20°C for warming (room temperature) alternations. Each cooling and warming was continued for two h. Hard seed was decreased as the treatment cycle was increased through the third cycle for Peace seeds (from 24 to 5%) and through the fifth cycle for Anik seeds (from 60 to 14%; Fig. 3). Germination was the greatest at the third cycle in Peace seeds (95%) and at the fifth cycle in Anik seeds (81%). Rutar et al. (2001) used the same treatment temperatures (-80°C for cooling and 20°C for thawing) and time (2 h) as the study of Stout (1990) in freeze-thaw scarification on alfalfa seeds, however the cooling device was dry ice and acetone instead of an ultra-low bio freezer. Dry ice and acetone cooled down seeds as a result of evaporation and these devices might not cool down all seeds equally as the freezer did. There was no influence of dry ice and acetone on hard seed or germination of alfalfa seeds.
Freeze-thaw scarification using liquid N (-196°C) showed varying results depending on the species. Seeds of Oxley CMV were soaked in liquid N for cooling (10 min) and then the seeds were soaked in steam bath at 100°C for warming (10 min) (Acharya et al., 1993). Germination of Oxley CMV seeds was increased from 0 in control to 86% by the freeze-thaw scarification when it was repeated for 15 times. In contrast, no influence was observed on germination or hard seed of milkvetch when seeds were scarified by freeze-thaw scarification with liquid N for cooling (five min) and room temperature for warming (Patane and Gresta, 2006).
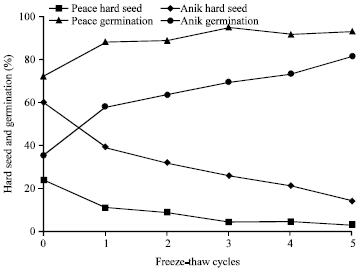 | |
| Fig. 3: | Hard seed and germination (%) of two cultivars of alfalfa exposed to freeze-thaw scarification with the temperature alternations between -80°C (2 h) and 20°C (2 h) (Stout, 1990) |
Freeze-thaw scarification with the use of liquid N had no influence on hard seed in 27 species, including ‘Grim’ alfalfa, ‘Chesapeake’ red clover, ‘Eski’ sainfoin (Onobrychis viciifolia Scop.), ‘A1564’ soybean (Glycine max L. Merr.) and ‘Purple’ common vetch (Vicia sativa L.) when soaked in liquid N for up to 180 days, while seeds of sesame (Sesamum indicum L.) and flax (Linum usitatissimum L.) were damaged by liquid N (Stanwood, 1980). Other studies showed increased abnormal seedlings by freeze-thaw scarification using liquid N. Seeds of rabbitfoot clover (T. arvense L.) were soaked in liquid N for cooling (5 min) and then placed at room temperature (slow warming) or hot water bath (40°C; rapid warming) for subsequent warming for 1 h (Pritchard et al., 1988). These treatments increased seedling abnormality but did not increase germination of the seeds as freeze-thaw cycle increased. Seedling abnormality of alfalfa (PI238151, PI258765, PI259522 and PI286378) was also increased when seeds were soaked in liquid N for 24 h (Wiesner et al., 1994). Freeze-thaw scarification using liquid N may have potential to reduce hard seed or improve germination (Acharya et al., 1993); however, in many instances, it had no influence on hard seed or germination (Stanwood, 1980; Pritchard et al., 1988; Wiesner et al., 1994; Patane and Gresta, 2006).
MECHANICAL SCARIFICATION
For mechanical scarification, many researchers have used mechanical scarifiers (Carleton et al., 1971; Townsend and Mcginnies, 1972; Miklas et al., 1987; Singh et al., 1991; Patane and Gresta, 2006; Dittus and Muir, 2010) or sandpapers to rub seeds manually (Baes et al., 2002; Uzun and Aydin, 2004; Patane and Gresta, 2006; Can et al., 2009).
A small vane-type scarifier has five rotation settings and scarification level is controlled by throwing seeds into the scarifier at different air pressures (Carleton et al., 1971). This scarifier increased germination of Lutana CMV from 39% in control to 60% when the seeds were thrown into the scarifier four times at 50 rpm, whereas rotation frequency higher than 50 (55 and 60 rpm) increased damaged seeds up to 36%. Another scarifier called Model No. 2 Foresberg Huller (Fig. 4) was made for larger seeds and it increased germination of Lutana CMV seeds from 20 in control to 78% when seeds were thrown into the scarifier four times at 1400 rpm (Carleton et al., 1971).
The other scarifiers introduced in the study of Carleton et al. (1971) were Model S scarifier and huller (Crippen Manufacturing Co., MI, USA) and Wes Gro process brand polisher (Northrup King Seed Co., Ontario, Canada).
 | |
| Fig. 4: | Model No. 2 Foresberg Huller (Can Seed Equipment Ltd. Available at: http://www.canseedequip.com/milling.php) |
These scarifiers had four rotation frequency settings, 700, 900, 1300 and 1600 rpm and have capability to scarify a large volume of seeds; however, both scarifiers had little influence on germination of Lutana CMV seeds as the highest germination obtained was only 53% (Carleton et al., 1971). On the other hand, a scarifier made and used by Townsend and Mcginnies (1972) successfully reduced hard seed of CMV seeds. This scarifier was a small drum type, in which abrasive paper was attached inside the drum. This was connected to air pressure that blew seeds into the drum by different level of air pressures. Among the three air pressures of 40, 60, 80 pound per square inch (psi) used in the study, 60 and 80 psi reduced hard seed from 40 in control to 2% when seeds were placed into the scarifier for two times (Townsend and Mcginnies, 1972). There were no differences in hard seed when seeds were scarified with 50 or 60 grits sandpapers attached inside the drum. Damaged seeds were increased with increased treatment times at 80 psi pressure.
The same scarifier was used in the study of Patane and Gresta (2006) to scarify seeds of button medic and milkvetch. Seeds were placed into the scarifier for 10 times. Little influence of this scarification was observed on hard seed reduction and improvement of germination. On the other hand, when seeds were rubbed manually with sandpapers (100 grits), initial hard seeds (average of the two species 94%) and germination (11%) of the two species became 1 and 92%, respectively. Mechanical scarification using sandpapers was also effective on other Medicago (M. hispida, M. arabica, M. scutellata, M. lupulina and M. falcata) and Trifolium species (T. resupinatum, T. subterraneum, T. alexandrinum, T. meneghinianum and T. striatum). Germination of these Medicago (initial 10-18%) and Trifolium species (initial 10-40%) were increased to 90-97% and 85-95% when rubbed with sandpapers, respectively (Uzun and Aydin, 2004). Effectiveness of sandpaper scarification was also reported on M. rigidula, M. rotata, M. orbicularis, M. scutellata and T. spumosum (Can et al., 2009). Seeds were manually shaken in a bottle in which sandpaper was installed inside. Germination of these Medicago species (initial 3-15%) and T. spumosum (initial 5%) was increased to 73-100% and 80%, respectively. Sandpaper scarification, however, did not affect seeds of M. scutellata, M. turbinate, T. striatum (Uzun and Aydin, 2004); T. cherleri, T. spadiceum, T. lappaceum, T. scabrum, T. angusrifolium, T. strictum and T. badium (Can et al., 2009). It appears that mechanical scarification is effective on Medicago (Uzun and Aydin, 2004; Patane and Gresta, 2006; Can et al., 2009), Trifolium (Uzun and Aydin, 2004; Can et al., 2009) and Astragalus (Patane and Gresta, 2006); however the effectiveness of mechanical scarification may vary depending on the genus and species (Uzun and Aydin, 2004; Can et al., 2009).
ACID SCARIFICATION
Acid scarification is considered as one of the most effective scarification methods used for seed scarification. Sulfuric acid is the most popular and effective chemical to reduce hard seed of legume seeds (Pandrangi et al., 2003; Martin and De La Cuadra, 2004; Uzun and Aydin, 2004; Patane and Gresta, 2006; Can et al., 2009). The effectiveness of acid scarification depends on concentration of acid, duration of scarification and species and cultivars used (Martin and De La Cuadra, 2004; Alderete-Chavez et al., 2011).
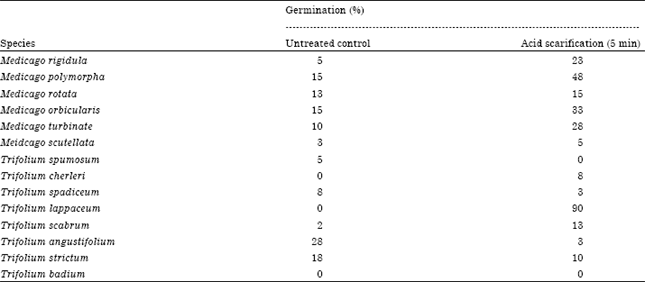
Many researchers used acid scarification in Medicago and Trifolium species. For example, seeds of six Medicago (M. rigidula, M. polymorpha, M. rotata, M. orbicularis, M. turbinata and M. scutellata) and eight Trifolium species (T. spumosum, T. cherleri, T. spadiceum, T. lappaceum, T. scabrum, T. angustifolium, T. strictum and T. badium) were soaked in sulfuric acid (95-97%) for five min (Can et al., 2009). Germination of T. lappaceum was increased from 0 in untreated control to 90%; however, no or little changes were observed in rest of the Medicago and Trifolium species (Table 1). Other studies also showed that acid scarification for five min has no effect on seeds of M. orbicularis (Crawford, 1976; Russi et al., 1992a, b) and M. scutellata (Uzun and Aydin, 2004). Acid scarification was also not effective in alfalfa seeds. Pandrangi et al. (2003) scarified seeds of alfalfa with various concentrations of sulfuric acid (0.1, 0.2, 0.5, 1.0, or 2.0 N) under several treatment timings (2.5, 5, 10, 15, 20, 30, 45 and 60 min).
| Table 1: | Effect of acid scarification on Medicago and Trifolium species (Can et al., 2009) |
 | |
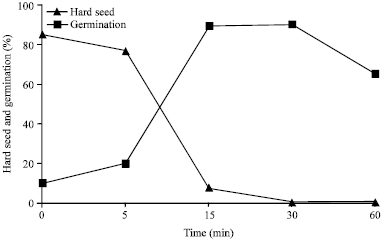 | |
| Fig. 5: | Hard seed and germination of M. polymorpha seeds treated with concentrated sulfuric acid (Martin and De La Cuadra, 2004) |
All concentrations had little influence on germination of alfalfa seeds (range 85-95%) and reduction of germination (range 74-76%) occurred when seeds were soaked longer than 45 min. Acid scarification was not also effective on seeds of milkvetch and button medic when three concentrations of sulfuric acid (30, 50 and 70%) were used for 15, 25, 35 and 60 min (Patane and Gresta, 2006).
In contrast, (Martin and De La Cuadra, 2004) reported that the hard seed of M. polymorpha could be successfully reduced by acid treating the seeds for 15 min. In this study, there was a significant reduction in hard seed (from 84 to 7%) and increase in germination (from 10 to 89%) in M. polymorpha (Fig. 5). Balouchi and Sanavy (2006) also showed the positive influence of sulfuric acid (96%) scarification on seeds of M. polymorpha and M. rigidula. Germination of M. polymorpha and M. rigidula was increased from 12 and 13% in untreated control to 96% after treating with sulfuric acid for 10 min. Germination of five cultivars of alfalfa seeds was shown to be increased in another study from 45 in control to 85% when seeds were soaked in concentrated sulfuric acid for 30 min (Tomer and Maguire, 1989). Germination of Chinese milkvetch (A. sinicus L.) seeds was also improved from 8 in untreated control to 93% when soaked them in concentrated sulfuric acid for 20 min (Kim et al., 2008). The positive effect of acid scarification was reported for other forage legumes, such as Lupinus angustifolius (Burns, 1959), Centrosema pubescens (Pe et al., 1975), Vigna mungo (Tomer and Kumari, 1991), V. umbellate (Tomer and Singh, 1993), Ornithopus compressus and O. pinnatus (Fu et al., 1996), Trigonella corniculata (Pandita et al., 1999) and Tamarindus indica (El-Siddig et al., 2001), L. leptophyllus (Alderete-Chavez et al., 2010a), Crotalaria retusa (Alderete-Chavez et al., 2010b).
Variable successes have been reported by many researchers for different species and even cultivars within the same species. For example, acid scarification had little influence on many Medicago (Crawford, 1976; Russi et al., 1992a, b; Pandrangi et al., 2003; Martin and De La Cuadra, 2004; Uzun and Aydin, 2004; Can et al., 2009), Trifolium (Can et al., 2009); Astragalus species (Patane and Gresta, 2006), whereas it was effective in seeds of M. polymorpha (Martin and De La Cuadra, 2004; Balouchi and Sanavy, 2006), M. rigidula (Balouchi and Sanavy, 2006), T. lappaceum (Can et al., 2009) and several cultivars of alfalfa when seeds were treated with 95-97% sulfuric acid for 5, 15 and 30 min, respectively (Tomer and Maguire, 1989). These variable results by acid scarification may be due to the composition change in seed coat.
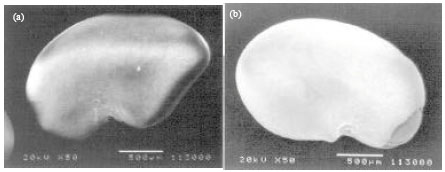 | |
| Fig. 6(a-b): | Alfalfa seeds under microscope. Untreated alfalfa seed (a) shows waxy coating and no damage, (b) while acid treated (sulfuric acid) seed shows no waxy coating. Acid treatment has removed and damaged seed coat (Pandrangi et al., 2003) |
Acid scarification might have removed the thick palisade layer of seed coat (Fig. 2). The removal of the hard seed coat by acid scarification was shown in alfalfa seeds (Pandrangi et al., 2003; Fig. 6). The untreated alfalfa seed had waxy coating while acid scarified seeds had none. Effectiveness of the acid scarification may vary within the cultivars of same species, depending on their seed coat structures (Fairey and Lefkovitch, 1991; Taia, 2004; Patane and Gresta, 2006).
Forage legumes are highly valuable crops because of their ability to fix atmospheric N and reduce fertilizer input cost and produce high quality forage. Hard seed in many of the forage legumes is one of the major constraints, however, for successful stand establishment. The hard seeds lower germination rate creating less competitive stands against weeds over resources (e.g., water, light and nutrients) in the establishment year (Dittus and Muir, 2010). Scarification methods such as heat, freeze-thaw, mechanical and acid scarification are useful tools to soften hard seeds, improve germination and enhance seedling establishment. However, effectiveness of the methods varies depending on the duration of imposed treatments and species or cultivars to be used (Taia, 2004). Over treatment or longer time scarification may impose negative impacts on or injury to the seeds. This review suggests that there is no single method of scarification that can be recommended for all legume species in general. As effectiveness of scarification methods varies among species and even cultivars within the same species, detailed scarification studies on legume seeds, especially on newly released cultivars, are necessary.