Research Article
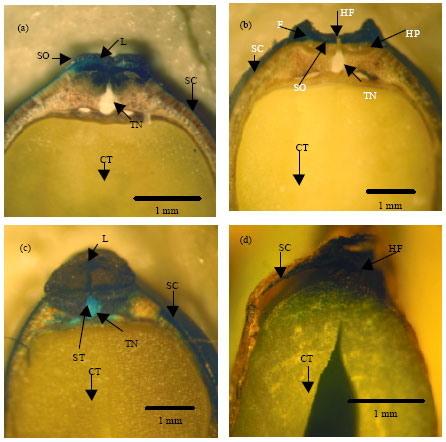
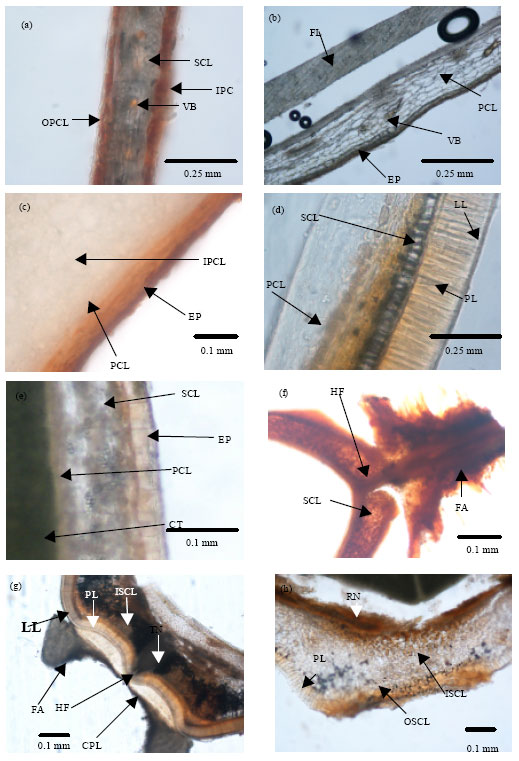
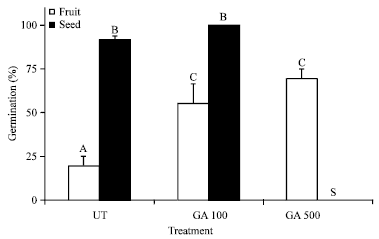
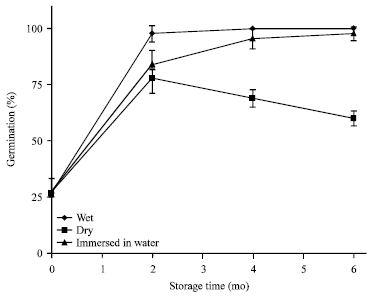
Variation in Seed Dormancy and Storage Behavior of Three Liana Species of Derris (Fabaceae, Faboideae) in Sri Lanka and Ecological Implications
Department of Botany, University of Peradeniya, Peradeniya, Sri Lanka
Jerry M. Baskin
Department of Biology, University of Kentucky, Lexington, KY, USA
Carol C. Baskin
Department of Biology, University of Kentucky, Lexington, KY, USA
M. Thilina R. Fernando
National Herbarium, Royal Botanic Gardens, Peradeniya, Sri Lanka










Dr.P.k.Nagar Reply
As such paper is good but the authors should have conducted some more physiol studies like moisture content ABA and related dormancy stidies. I would suggest that the email addrss of corresponding author may also b given