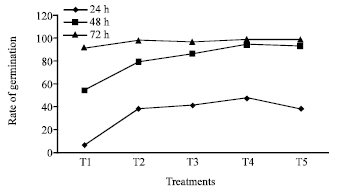
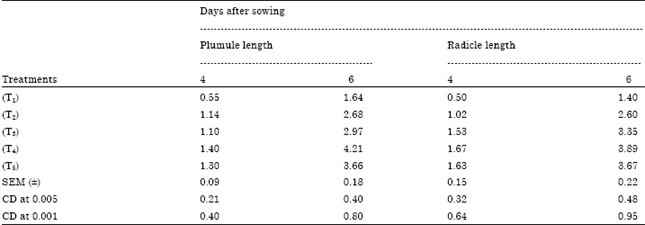
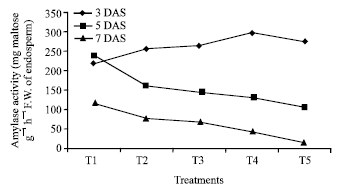
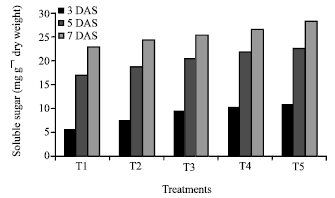
Research Article
Role of Seed Hardening in Rice Variety Swarna (MTU 7029)
Department of Plant Physiology, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, India
P. Vijai
Department of Plant Physiology, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, India
B. Bose
Department of Plant Physiology, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, India