Research Article
Bioactive Non-alkaloidal Secondary Metabolites of Hyoscyamus niger Linn. Seeds: A Review
Pharmacy Group, Birla Institute of Technology and Science-Pilani, Hyderabad Campus, Jawahar Nagar, Hyderabad 500 078, India
In view of the impact of plant secondary metabolites on human fitness, there has been an upsurge in interest for phytochemical investigations to discover novel pharmacologically active drugs and fine chemical substances from plants. The well known anticholinergic alkaloid rich plant, Hyoscyamus niger Linn. (Duke, 1985) has been a subject of study by many multidisciplinary scientists for its various bioactive principles possessing potent therapeutic properties. H. niger belonging to solanaceae family is commonly known as Henbane or Hogs bean in English, Parasigaya in Sanskrit and Khursani ajwayan in Hindi (Kirtikar and Basu, 1975).
The alkaloidal contents of H. niger are well documented, thereby Egyptian henbane is found to possess greater percentage of alkaloids (0.7-1.5%) than European henbane. It originally contains optically active levorotatory alkaloid, l-hyoscyamine as major alkaloid. The racemic form of l-hyoscyamine, atropine and scopolamine are often found in leaves (Robbers et al., 1996), while the root part is reported to contain apoatropine (atropamine) and cuscohygrine (Haga, 1954). Henbane seeds contain about 0.06-0.1% of alkaloids like hyoscyamine with a little hyoscine and atropine (Uniyal, 1989). Besides these tropane alkaloids, seven nortropane alkaloids known as calystegins are also reported from seeds of H. niger (Naoki et al., 1996). An extensive review on the existence of various secondary metabolites of seeds of H. niger has elaborated the presence of number of non-alkaloidal constituents along with alkaloids. The present study discusses the scientific information on the non-alkaloidal secondary metabolites of H. niger seeds.
MORPHOLOGY AND DISTRIBUTION
H. niger, a greyish green shrub consists of leaves and stem with flowering tops giving strong unpleasant smell and bitter taste. Two forms of this species, biennial and annual are known and used medicinally. The scented flowers are hermaphrodite and are pollinated by insects (Melchers, 1939). A single plant yields around 10,000 seeds/harvest. Henbane seeds are dark grey in color, reniform in shape and about 1.5 mm long having a minutely reticulated testa (Uniyal, 1989).
It is distributed throughout Europe i.e., from Portugal to Greece in south and from Norway to Finland in north, Caucasia, Iran, Asia, North America and Serbia. In India, it is generally found in Jammu and Kashmir (Garhwal Himalayas, 8000-11000 feet high), Himachal Pradesh and Kumaon of Uttar Pradesh (Duke, 1985).
ETHNOPHARMACOLOGICAL USES
H. niger, though recorded as a poisonous plant, is traditionally used in Indian as well as Chinese medicine for its use in stomach cramps, heavy coughs, neuralgia and manic psychosis. The plant is also said to possess anti-spasmodic, sedative and analgesic properties (Duke, 1985). In Tibetan medicine, the seeds of H. niger are used as anthelmintic, antitumor and febrifuge. They are also found to be useful in the treatment of stomach/intestinal pain due to worm infestation, toothache, infection of pulmonary regions and tumors (Tsewang, 1994). It is extensively used as a pain killer when affecting the urinary tract, especially when suffering from kidney-stone. Externally the seed oil is used for neuralgic, dental and rheumatic pains (Bown, 1995; Chevallier, 1996).
NON-ALKALOIDAL SECONDARY METABOLITES
While the poisonous henbane is well known for its alkaloidal content, the occurrence of non-alkaloidal metabolic constituents is not well explored. The anticholinergic plant species also produces non-alkaloidal secondary metabolites like withanolides, flavonoids, lignans, coumarinolignans, saponins, glycerides, glycosides, phenolics etc. (Table 1). The elaborated pharmacological activities of H. niger could be attributed to its wealthy chemical constituents.
| Table 1: | List of non-alkaloidal constituents isolated from Hyoscyamus niger seeds |
 | |
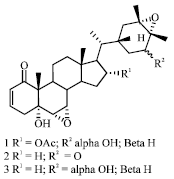 | |
| Fig. 1: | Structure of withanolides isolated from H. niger seeds |
Withanolides
Withanolides are a group of naturally occurring steroidal lactones i.e., 22-hydroxyergostan-26-oic acid-26,22-lactone possessing interesting biological activities like antitumor, anti-inflammatory, antimicrobial and brain regenerative properties. Withanolides are found to occur mainly in solanaceous plants. Ma et al. (1999) first reported the existence of withanolide class steroids in the seeds of H. niger. Withanolides: Daturalactone-4 (2), hyoscyamilactol (3), 16α-acetoxyhyoscyamilactol (1) were isolated and characterized from the ethanolic extract of seeds (Fig. 1).
Lignans
With the enduring interest in understanding the non-alkaloidal principles of seeds of H. niger, Begum et al. (2006a) reported the isolation of a novel lignan, hyosmin (4) and characterized it as 3-{[(2R)-2-carbomethoxy-2-hydroxy]ethyl}benzoate ester of {(2R,3S,4S)-2-(4-hydroxy-3-methoxyphenyl)-3-hydroxymethyl-4-[(4-hydroxy-3-methoxyphenyl)methyl] tetrahydrofuran. Further, the same group isolated and reported two more new lignans of tetrahydrofurano type, named as hyoscyamal (5) and balonophonin (6) (Begum et al., 2009). Lignans are a biologically active group of natural products formed by the union of two phenylpropanoids i.e., C6C3 units by oxidative coupling. Reviewing the literature, it is found that the biosynthesis of lignans in H. niger effected with the formation of new group of metabolite called lignanamides. Ma et al. (2002) isolated and elucidated the structure of four such lignanamides, hyoscyamide (7), grossamide (8), cannabisin D (9) and cannabisin G (10) from the seeds (Fig. 2).
Coumarinolignans
The continued phytochemical investigation in searching the non-alkaloids led by Begum et al. (2006b) culminated with the identification of presence of a new group of compounds called coumarinolignans in the seeds of H. niger. Coumarinolignans are the structural combination of coumarins and phenylpropanoids, bioderived by the oxidative coupling process. Five new coumarinolignans (Fig. 3), cleomiscosin A (11), cleomiscosin B (12), cleomiscosin A-9'-acetate (13), cleomiscosin B-9'-acetate (14) (Begum et al., 2006a) and cleomiscoin A methyl ether (15) were isolated from the methanolic extract through fractionation and chromatographic separation (Begum et al., 2010). Amongst solanaceous plants, H. niger is the first and only source identified to possess coumarinolignans.
Flavonoids and Flavonoid Glycosides
Phytochemical reports explore the occurrence of flavonoids like rutin, spiraeoside and 3',5-dihydroxy-3,4',5',6,7-pentamethoxyflavone in the seeds of H. niger (Begum et al., 2006b; Steinegger and Sonanini, 1960).
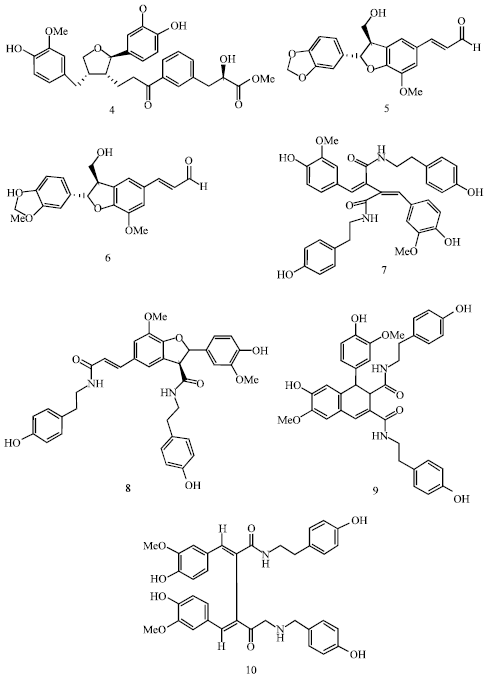 | |
| Fig. 2: | Structure of lignans and lignanamides isolated from H. niger seeds |
Further the existence of a furanoflavonoid glucoside, pongamoside C (16) and a flavonol glucoside, pongamoside D (17) were explicated (Fig. 4) by Begum et al. (2009).
Saponins
The seeds of black henbane had been identified as a new source for steroidal saponins (hyoscyamozides) of spirostan, furostan and pregnan series (Lunga et al., 2008). Hyoscyamozides A (18), B (19), B1 (20), B2 (21), B3 (22), C (23), Ñ1 (24), C2 (25), D, D1, Å (26), E1, F, F1 (27), J and J1 have been isolated and they are found to differ in the structure of aglycone and oligosaccharide chain (Fig. 5). While hyoscyamozides A, B, B1, B2, C, C1, D and D1 are derivatives of tigogenin, hyoscyamozide A3 is that of diosgenin.
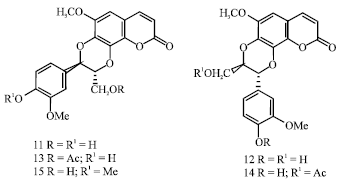 | |
| Fig. 3: | Structure of coumarinolignans reported from H. niger seeds |
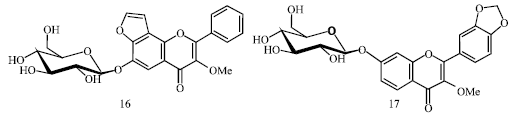 | |
| Fig. 4: | Structure of pongamoside C and pongamoside D |
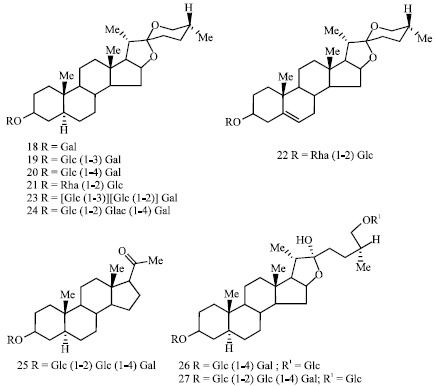 | |
| Fig. 5: | Structure of steroidal glycosides reported from H. niger seeds |
Furostan glycosides, hyoscyamozides E, E1, F, F1, J and J1 are derivatives of (25R)-5-furostan-3,22,26-triol and that of E2 of (25R)-5-furosten-3,22,26-triol.
Miscellaneous
H. niger seeds were also reported to contain steroidal glycosides (atroposide A, atroposide C, atroposide E, petunioside L), phenolics (vanillic acid, vanillin, pinoresinol and N-trans-feruloyl tyramine) and phytosterols (daucosterol, β-sitosterol), etc. (Begum et al., 2006b; Ma et al., 2002).
Biological Activities
Although, a large number of non-alkaloids have been isolated from H. niger, only a few have been studied for the biological activity. Lignanamides isolated from the seeds of H. niger were screened for cytotoxicity using human prostate cancer LNCaP cells. Grossamide (IC50 = 33 μM), cannabisin G (IC50 = 76 μM), cannabisin D (IC50 = 81 μM), displayed low levels of inhibitory activity (Ma et al., 2002). Evaluation of the analgesic, anti-inflammatory and antipyretic activities of the methanolic extract of seeds of H. niger had shown significant activity. The major non-alkaloidal constituent, cleomiscosin A was found to show anti-inflammatory activity under cotton pellet granuloma assay in mice. But its regioisomer cleomiscosin B was found to be inactive (Begum et al., 2010) under the study.
The antimicrobial activity of non-alkaloidal extract of seeds of H. niger were tested against Gram positive and Gram negative bacteria and a fungus (Khan et al., 1992). The effect was found to be potent against Aspergillus niger, Bacillus subtilis, Escherichia coli and Staphylococcus aureus.
The crude extract of H. niger has been studied by Khan and Gilani (2008) for hypotensive, cardiosuppressant and vasodilator activities. The extract was found to show a dose dependant (10-100 mg kg-1) fall in arterial blood pressure through a Ca2+-antagonist mechanism. To rationalize some of the medicinal uses of Hyoscyamus niger, Gilani et al., (2008) further studied the spasmolytic, antidiarrhoel, antisecretory, bronchodialatory and urinary relaxant properties. This experimental study showed that the crude extract of H. niger caused a complete concentration-dependent relaxation of spontaneous contractions of rabbit jejunum through a Ca2+ channel-blocking mechanism in addition to an anticholinergic effect. H. niger crude extract exhibited antidiarrhoeal and antisecretory effects against castor oil-induced diarrhoea and intestinal fluid accumulation in mice. The antispasmodic effect of H. niger was found to be mediated through a combination of anticholinergic and Ca2+ antagonist mechanisms (Gilani et al., 2008).
In a study made by Reza et al. (2009) the effect of methanolic extract of Hyoscyamus niger L. on the seizure induced by picrotoxin in mice was evaluated. The latency of seizure was increased with the pretreatment of 100, 200, 300 and 400 mg kg-1 of extract, thus showing the anticonvulsant activity against picrotoxin-induced seizures.
Throughout the literature review, one can see that, generally, the seeds of Hyoscyamus niger have been scientifically well evaluated. This review has explored that though alkaloids are the common secondary metabolites of solanaceous plants, there are a large number of non-alkaloidal constituents that include lignans, coumarinolignans, withanolides, lignanamides, glycerides, saponins, flavonoids and its glycosides, etc possessing various pharmacological activities. In conclusion, there are few studies about the pharmacological activity of non-alkaloidal constituents. Most of studies to date have focused their attention on the effect of crude extract of seeds. There is a deplorable scarcity of detailed pharmacological studies of chemical constituents published. Many of these chemical constituents could be biologically active and emerge as natural mimics, further as a dynamic natural formulation through systematic research. It is hopeful that this review may prompt many scientists and phytopharmaceuticals to undertake research in the area of non-alkaloids of solanaceous plants especially on H. niger.
Sincere thanks are due to Prof. M. Sahai for his suggestions and fruitful discussions in this work.