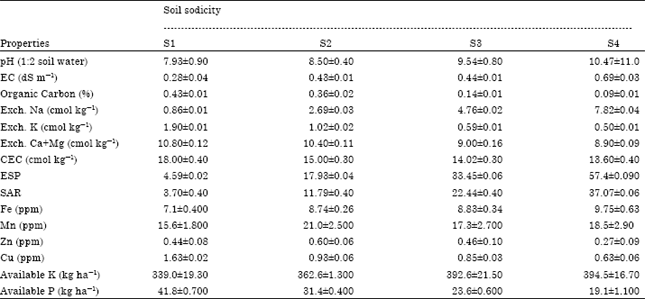
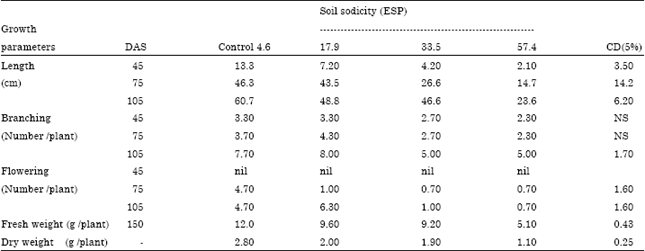
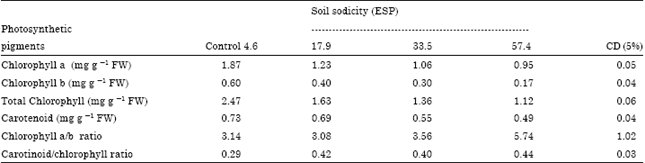
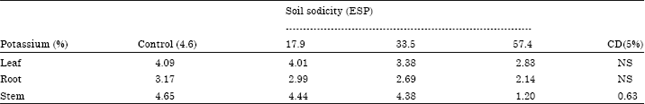
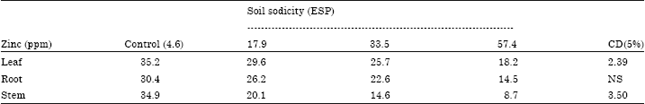
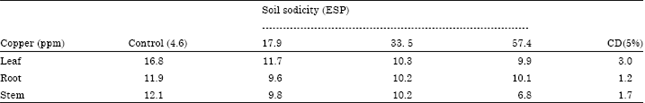
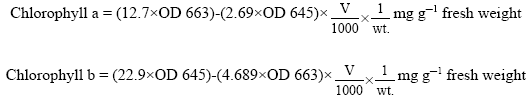Case Study
Effects of soil sodicity on growth, nutrients uptake and Bio-chemical responses of Ammi majus L.
University of Lucknow, Lucknow, Uttar Pradesh, India
Satyendra Tripathi
Department of Civil Engineering, Birla Institute of Technology and Science, Pilani, Hyderabad Campus, 500078, India
S.N. Pandey
University of Lucknow, Lucknow, Uttar Pradesh, India