Research Article
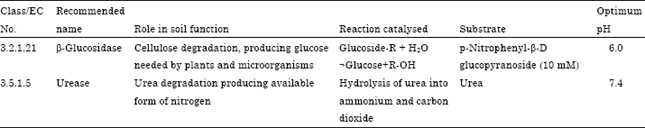
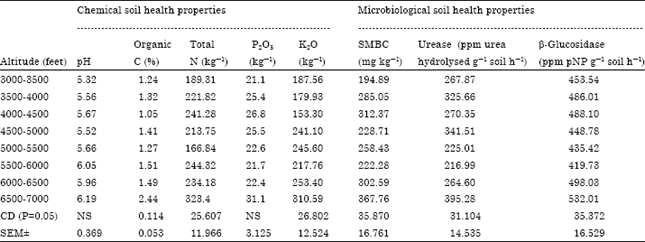
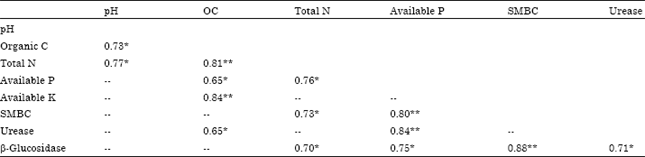
Influence of Altitudes on Activity of Soil Health Bioindicators β-glucosidase and Urease in Agricultural Soils of Almora District of Central Himalaya
Department of Soil Science and Agricultural Chemistry, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, India
Vimal Singh
Department of Soil Science and Agricultural Chemistry, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, India
P. P. Dhyani
GB Pant Institute of Himalayan Environment and Development, Kosi-Katarmal, Almora, India
Ashok Kumar
Department of Soil Science and Agricultural Chemistry, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, India