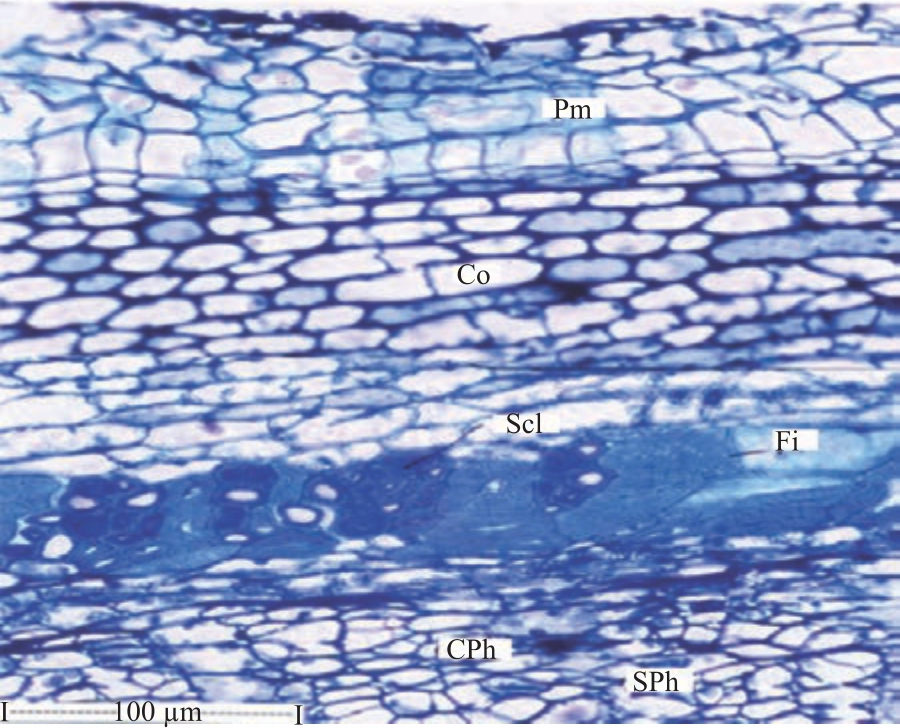
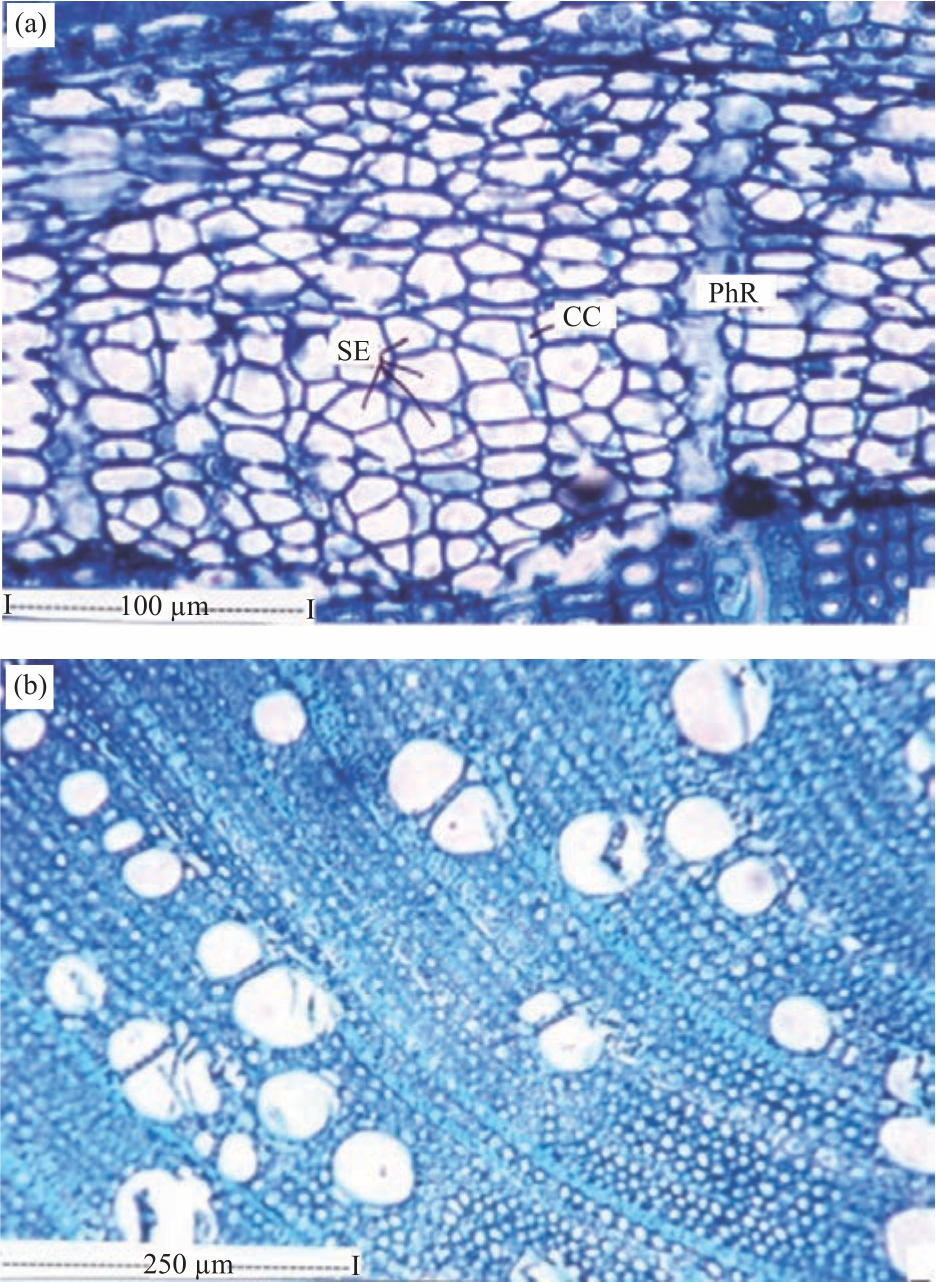
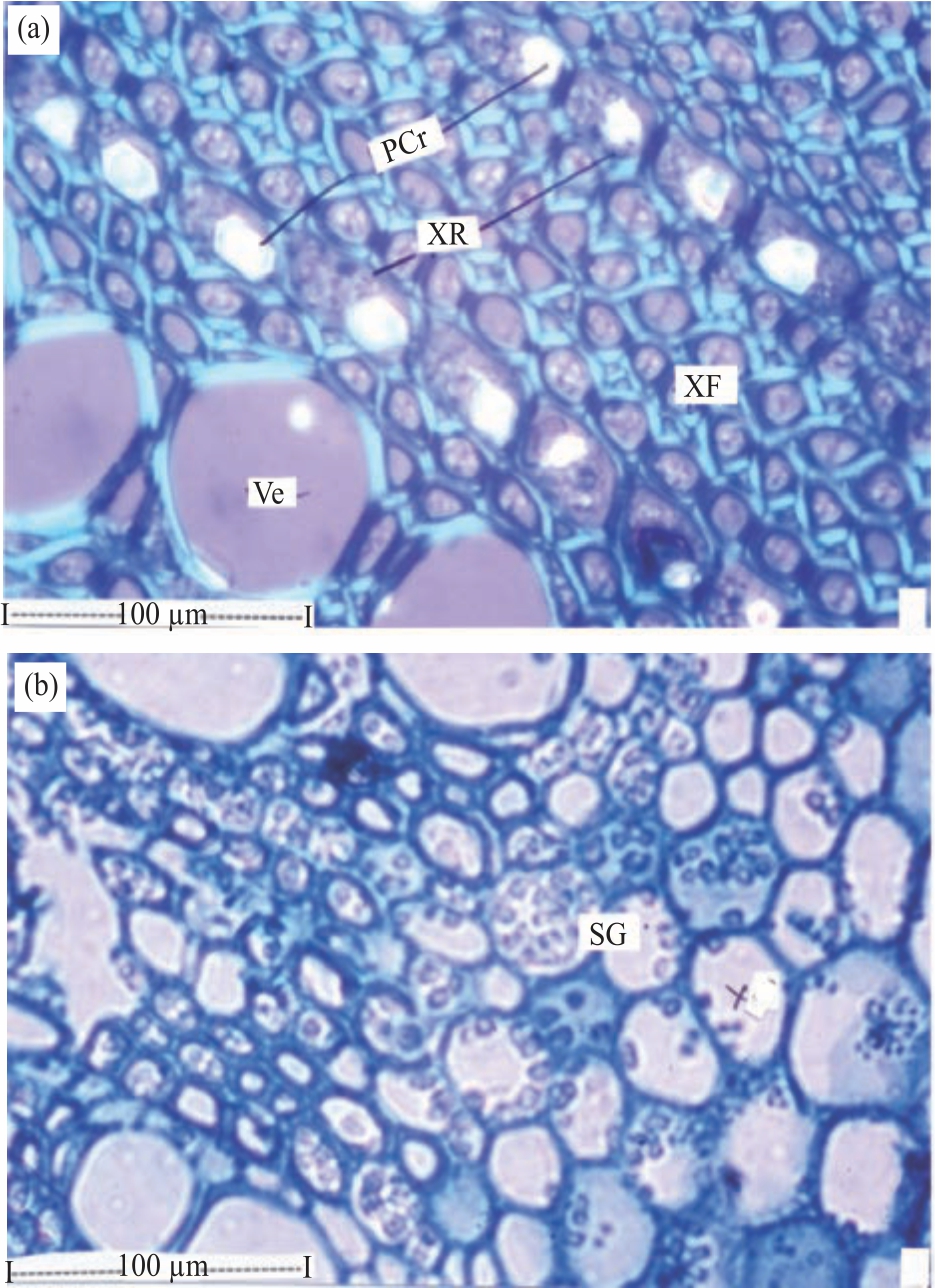
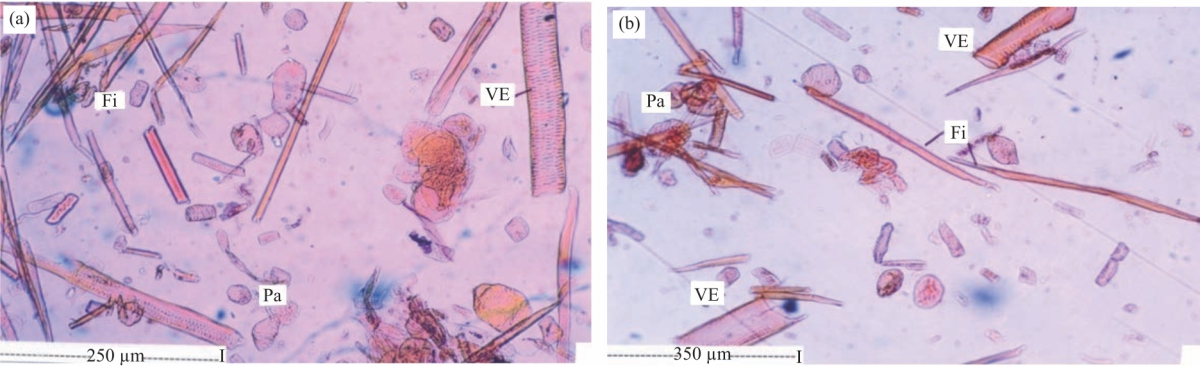
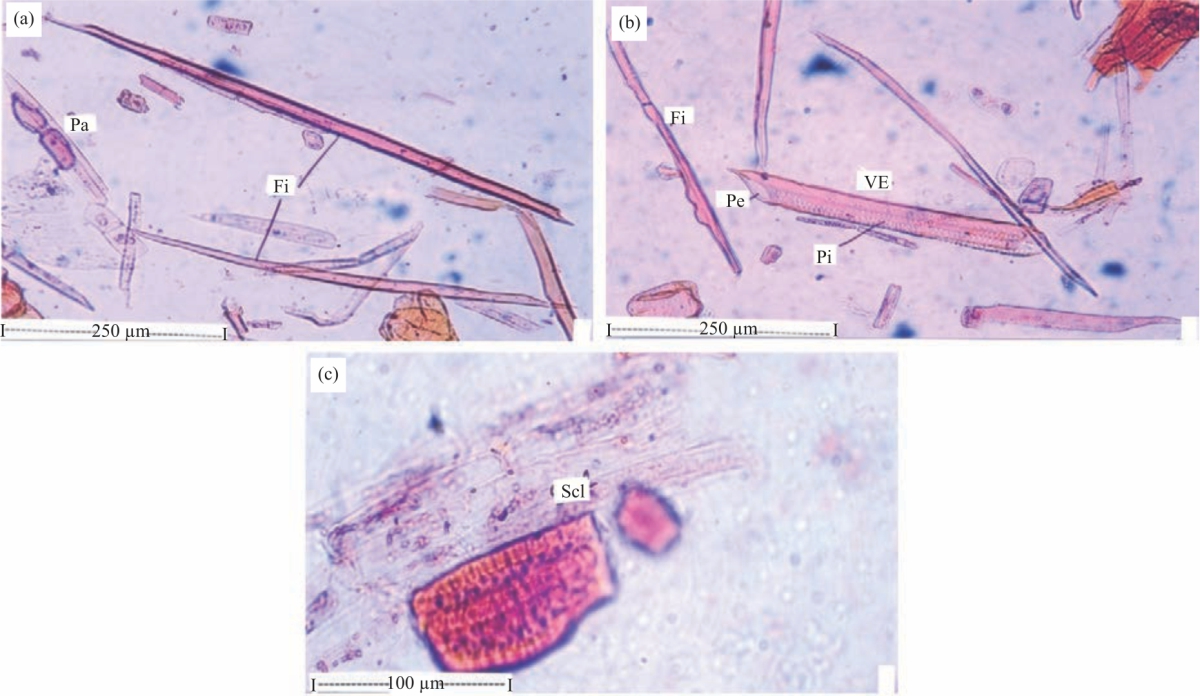
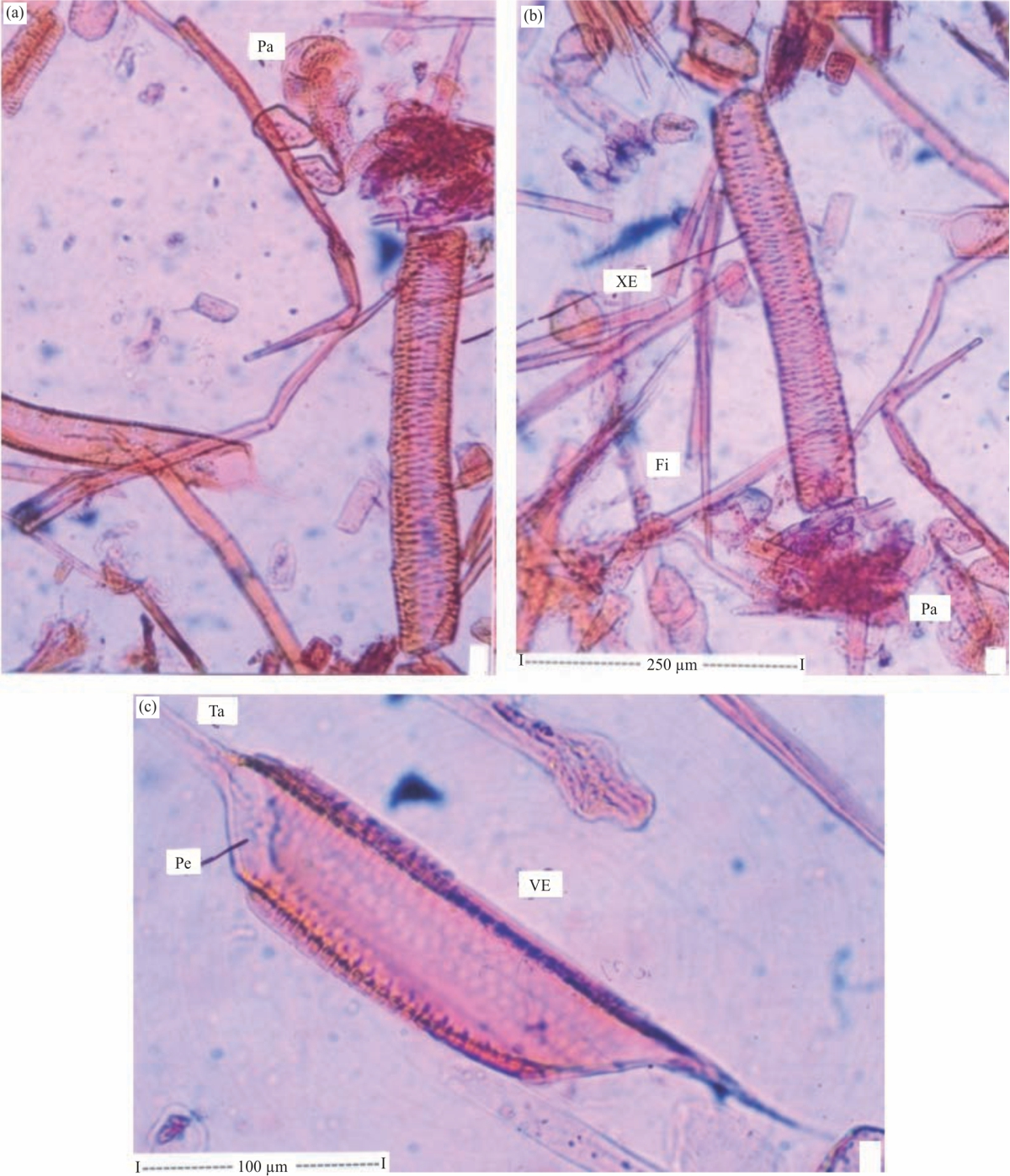
Research Article
Pharmacognostical and Phytochemical Analysis of Stems of Vitex pinnata Linn.
Department of Pharmacognosy, PGP College of Pharmaceutical Science and Research Institute, Namakkal (D.T), Tamil Nadu, India
Tarafaud Tahir
Faculty of Chemistry, Environmental Electrochemistry, Koya University, Koya, Iraq
Subasini Uthirapathy
Department of Pharmacology, Faculty of Pharmacy, Tishk International University, Kurdistan, Iraq`
LiveDNA: 91.35293