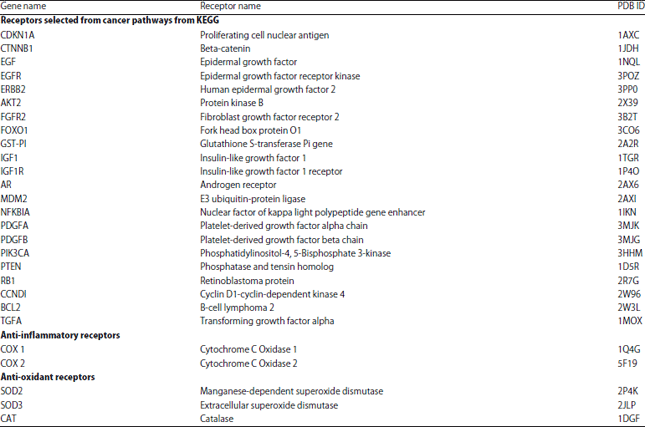
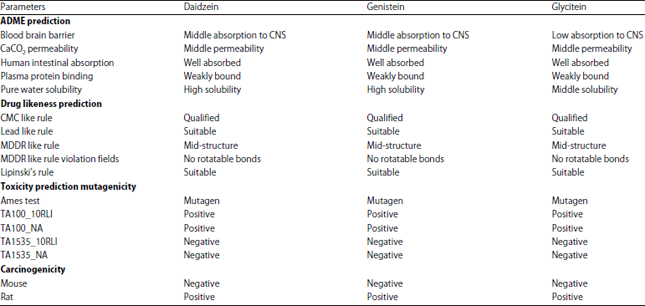
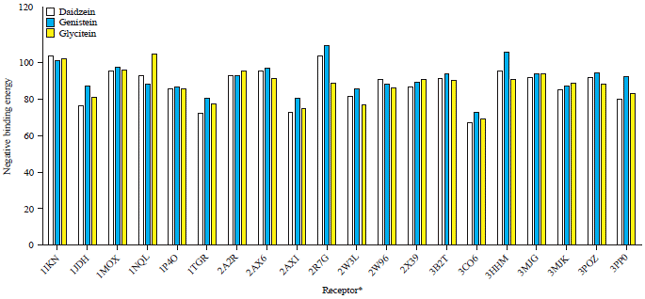
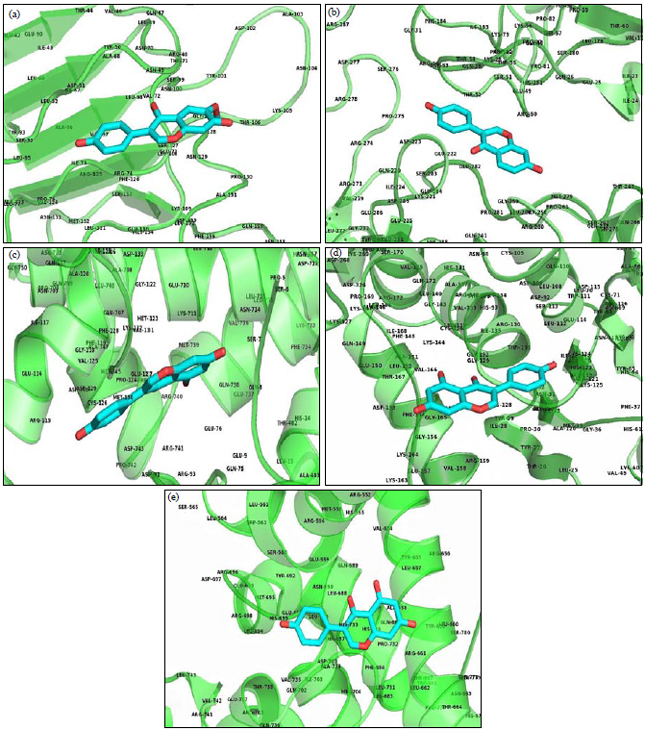
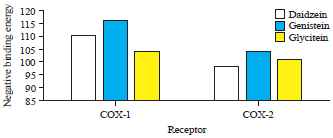
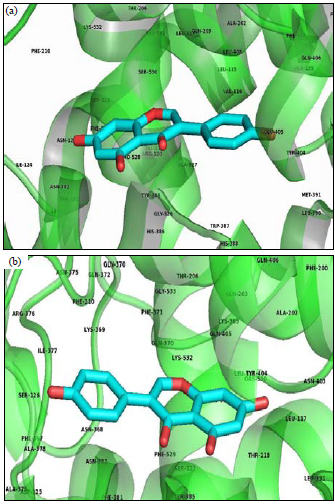
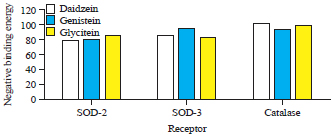
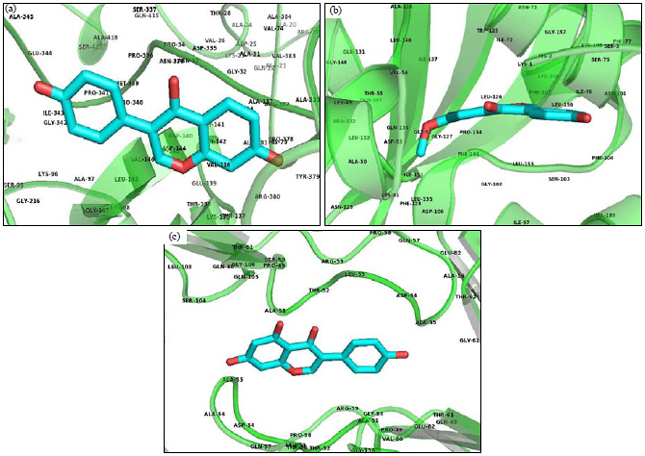
Research Article
Assessment of Anticancer, Anti-inflammatory and Antioxidant Properties of Isoflavones Present in Soybean
Dr. C.D. Sagar Center for Life Sciences, Dayananda Sagar College of Engineering, Visvesvaraya Technological University, Kumaraswamy Layout, 560 078 Bangalore, India
LiveDNA: 91.21334
Arpitha Mahajankatti
Dr. C.D. Sagar Center for Life Sciences, Dayananda Sagar College of Engineering, Visvesvaraya Technological University, Kumaraswamy Layout, 560 078 Bangalore, India
Yashas Devasurmutt
Dr. C.D. Sagar Center for Life Sciences, Dayananda Sagar College of Engineering, Visvesvaraya Technological University, Kumaraswamy Layout, 560 078 Bangalore, India
Ramakrishna Devappa
Dr. C.D. Sagar Center for Life Sciences, Dayananda Sagar College of Engineering, Visvesvaraya Technological University, Kumaraswamy Layout, 560 078 Bangalore, India
Kiran Sunderarajarao Vasist
Dr. C.D. Sagar Center for Life Sciences, Dayananda Sagar College of Engineering, Visvesvaraya Technological University, Kumaraswamy Layout, 560 078 Bangalore, India
Georgina Kosturkova
Bulgarian Academy of Sciences, Sofia, Bulgaria
Ravishankar Gokare Aswathanarayana
Dr. C.D. Sagar Center for Life Sciences, Dayananda Sagar College of Engineering, Visvesvaraya Technological University, Kumaraswamy Layout, 560 078 Bangalore, India