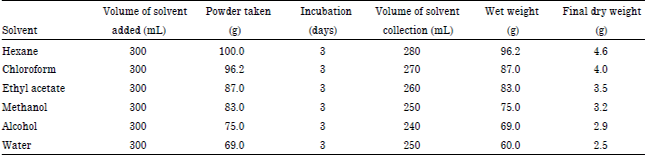
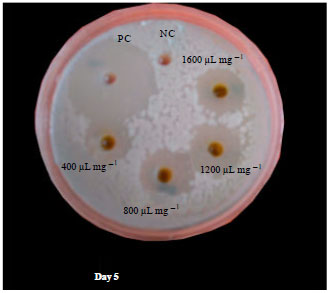
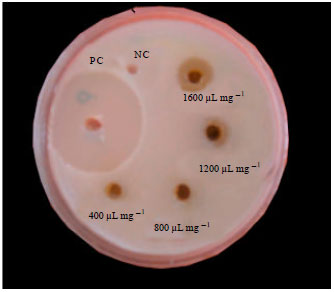
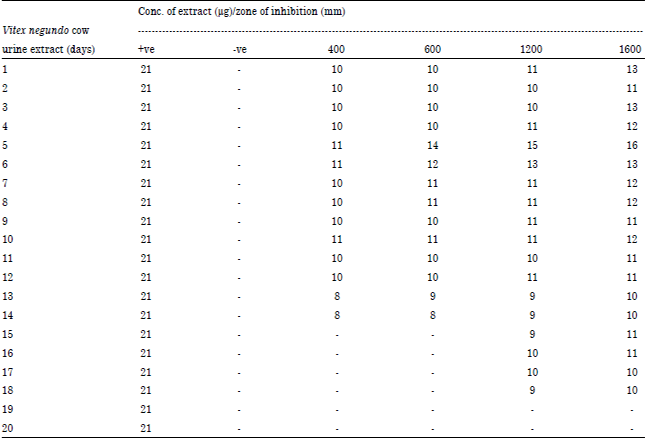
Research Article
Evaluation of the Antibacterial Potential, Preliminary Phytochemical Screening of Medicinal Plant against Plant Pathogen
Srimad Andavan Arts and Science College, T.V. Koil, Tiruchirapalli, 620020, Tamil Nadu, India
S. Pradeep Kumar
Department of Plant Science, Stress Physiology and Medicinal Plant Biotechnology, Bharathidasan University, Tiruchirappalli, 620024, Tamil Nadu, India