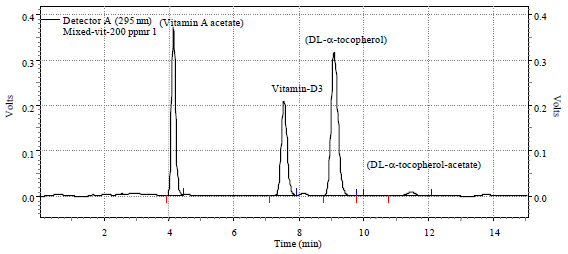
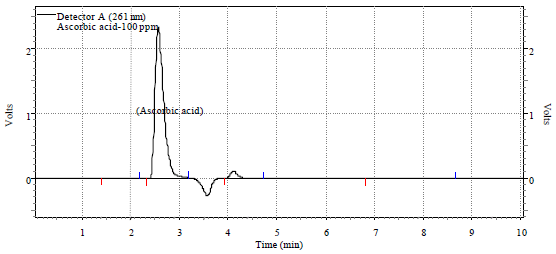
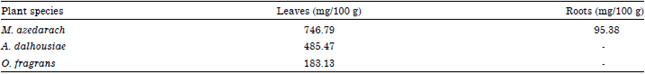
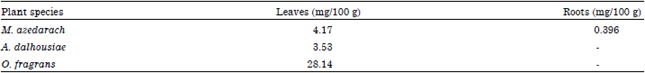Research Article
HPLC Analysis of α-tocopherol and Vitamin C in Important Medicinal Plants of Uttarakhand Himalayas
Department of Chemistry, Kumaun University, Nainital, 263002, Uttaranchal, India
S. Gupta
Department of Chemistry, Kumaun University, Nainital, 263002, Uttaranchal, India
G. Bisht
Department of Chemistry, Kumaun University, Nainital, 263002, Uttaranchal, India