Short Communication
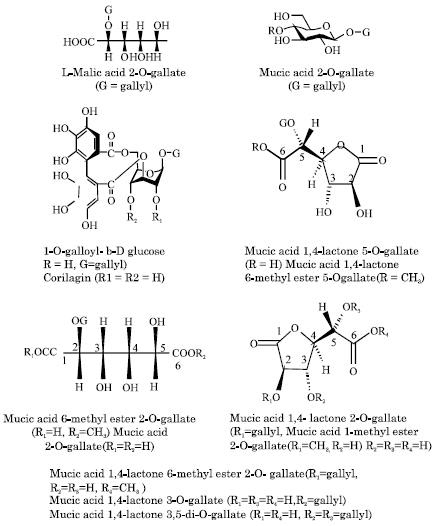
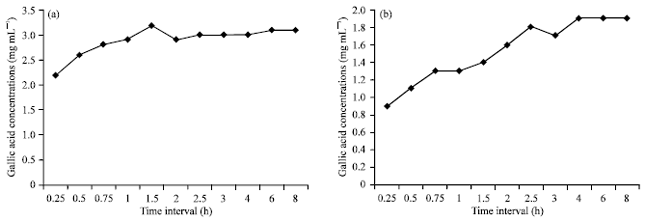
Biotransformation of Gallotannins From Fresh Fruit Juice of Emblica officinalis in In-vitro System
Department of Pharmacology, Institute of Pharmacy, Nirma University, Ahmedabad, 382 481, India
Ramesh K. Goyal
Department of Pharmacology, Narsee Monjee Institute of Management Studies, Mumbai, Maharashtra, 400 056, India