Review Article
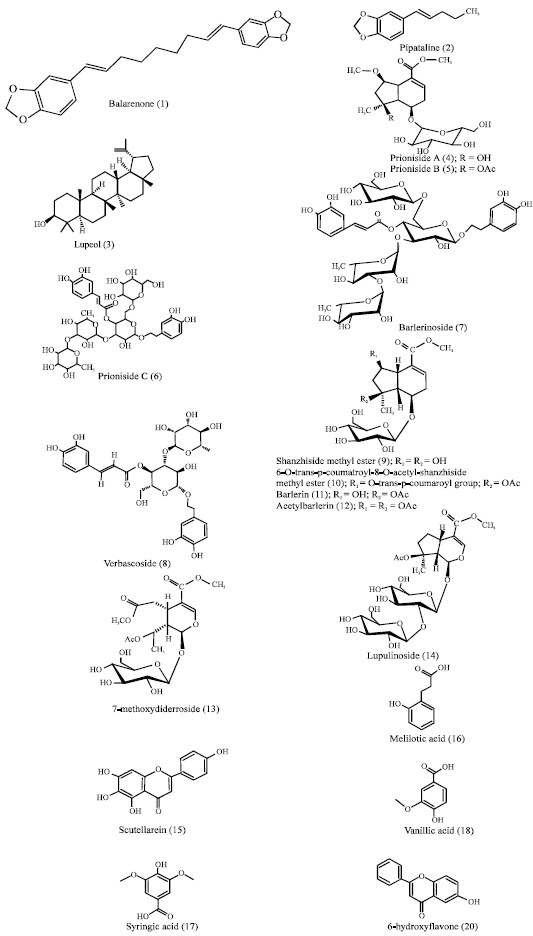
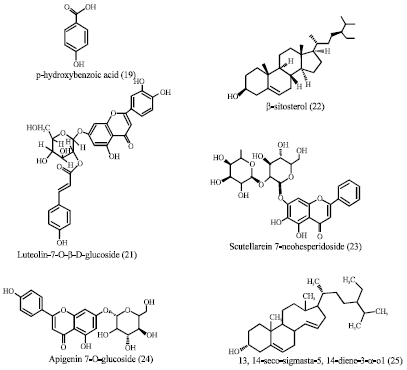
Barleria prionitis Linn.: A Review of its Traditional Uses, Phytochemistry, Pharmacology and Toxicity
Department of Botany and Forestry, Vidyasagar University, Midnapore (W)-721 102, India
A.K. Maji
Department of Botany and Forestry, Vidyasagar University, Midnapore (W)-721 102, India
S. Mahapatra
Department of Botany and Forestry, Vidyasagar University, Midnapore (W)-721 102, India
P. Banerji
Ulysses Research Foundation, Kolkata-700 029, India