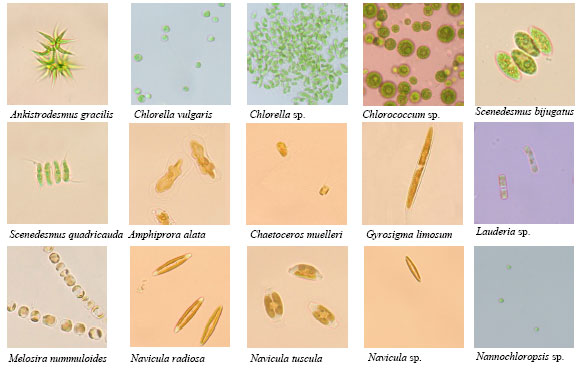
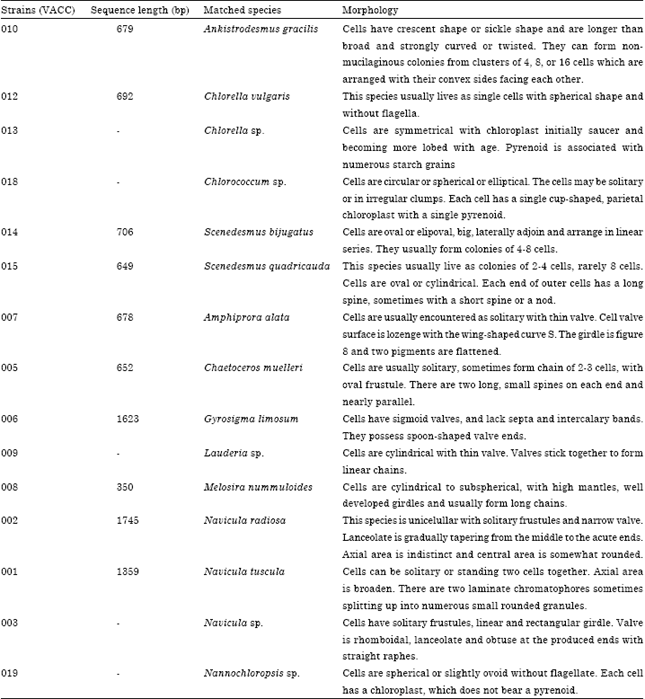
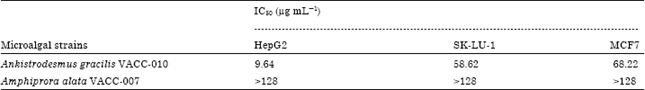
Research Article
Isolating and Screening Mangrove Microalgae for Anticancer Activity
Faculty of Biology, Hanoi National University of Education, Hanoi, Vietnam
D.N. Quang
Faculty of Chemistry, Hanoi National University of Education, Hanoi, Vietnam
N.T.H. Ha
Institute of Microbiology and Biotechnology, Vietnam National University, Hanoi, Vietnam
N.H. Tri
Faculty of Biology, Hanoi National University of Education, Hanoi, Vietnam