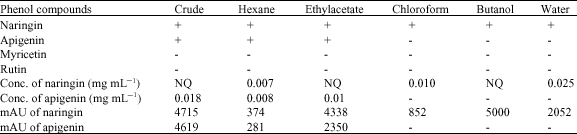
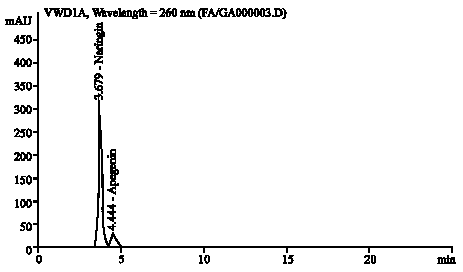
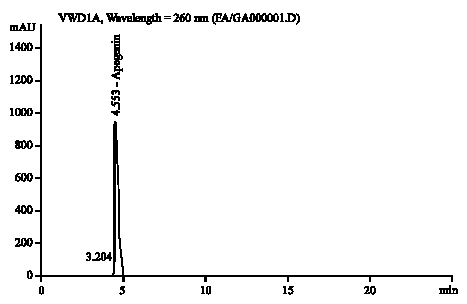
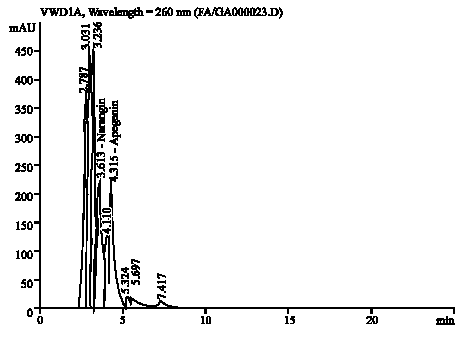
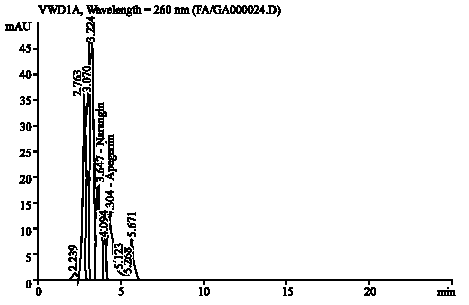
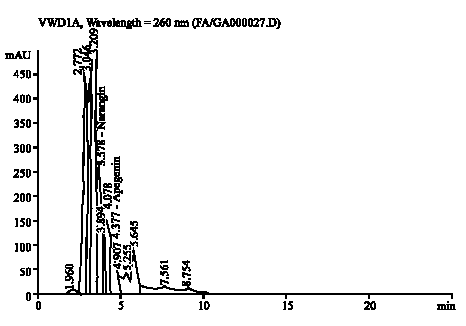
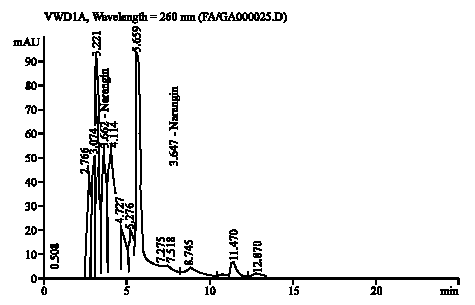
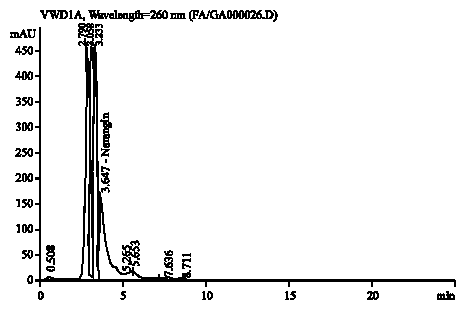
Research Article
In vitro Investigation of Antioxidant Phenolic Compounds in Extracts of Senna alata
Department of Cell Biology and Genetics, University of Lagos, Akoka, Lagos, Nigeria
H. Ogbunugafor
Department of Applied Biochemistry, Nnamdi Azikiwe University, Awka, Nigeria
G.K. Kareem
Department of Cell Biology and Genetics, University of Lagos, Akoka, Lagos, Nigeria
M.N. Igwo-Ezikpe
Department of Biochemistry, College of Medicine, Idi-Araba, Lagos, Nigeria