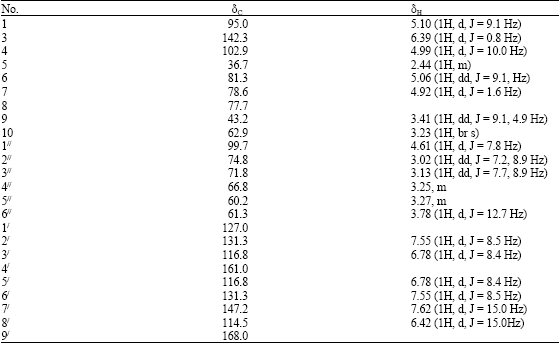
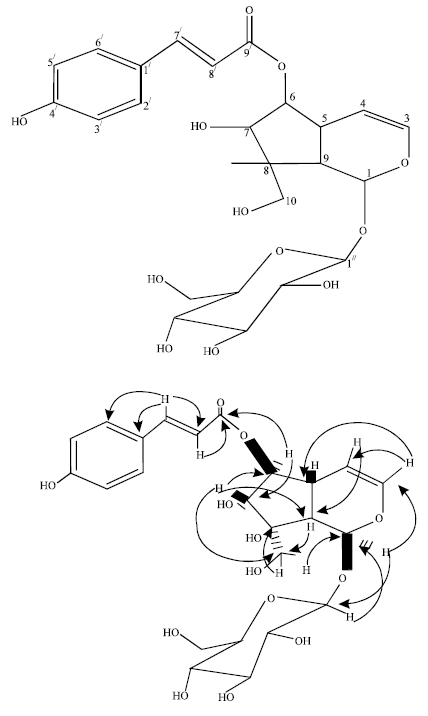
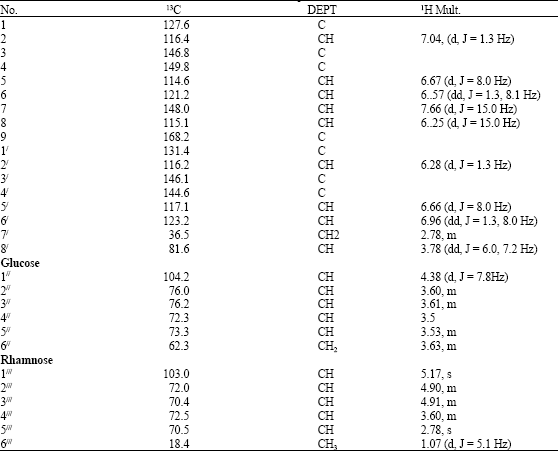
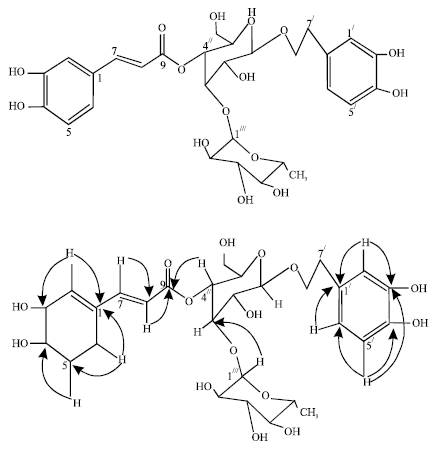
Research Article
Bioactive Chemical Constituents of Stereospermum kunthianum (Bignoniaceae)
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Benin, Benin City, Nigeria
I.M. Qadir
H.E.J. Research Institute of Chemistry, International Center for Chemical and Biological Sciences, University of Karachi, Karachi 75270, Pakistan
C.F. Poh
Department of Pharmacology, Faculty of Basic Medical Sciences, College of Health Sciences, Niger Delta University, Wilberforce Island, Yenagoa, Nigeria
K.I. Omogbai Eric
Department of Pharmacology and Toxicology, Faculty of Pharmacy, University of Benin, Benin City, Nigeria
M.I. Choudhary
H.E.J. Research Institute of Chemistry, International Center for Chemical and Biological Sciences, University of Karachi, Karachi 75270, Pakistan