Research Article
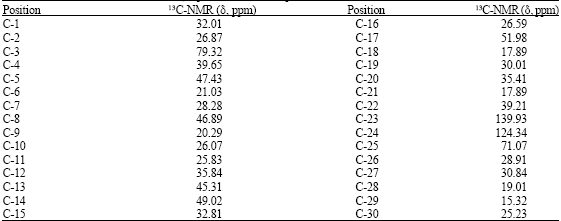
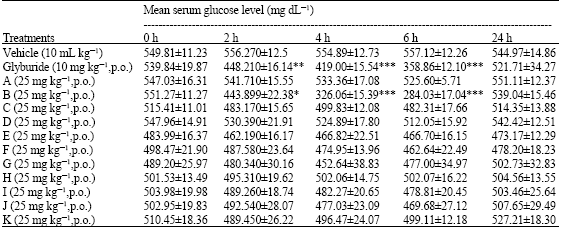
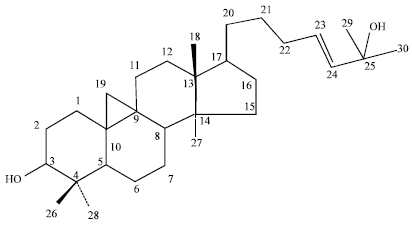
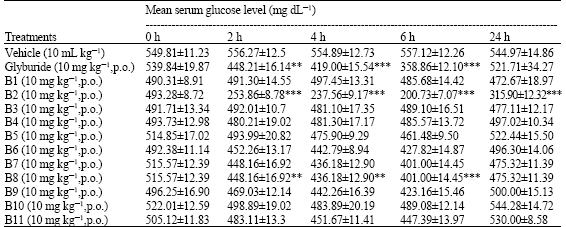
Antihyperglycaemic Activity of Cycloart-23-ene-3β, 25-diol Isolated from Stem Bark of Pongamia pinnata in Alloxan Induced Diabetic Mice
Department of Pharmacology, Poona College of Pharmacy, Bharati Vidyapeeth University, Paud Road, Erandwane, Pune-411 038, Maharashtra, India
Subhash L. Bodhankar
Department of Pharmacology, Poona College of Pharmacy, Bharati Vidyapeeth University, Paud Road, Erandwane, Pune-411 038, Maharashtra, India









vishal mali Reply
its very nice work in diabetes, new molecule development from plant origine for treatment of diabetes