Research Article
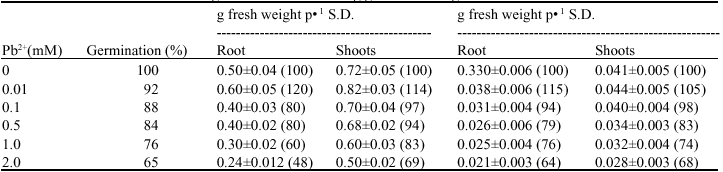
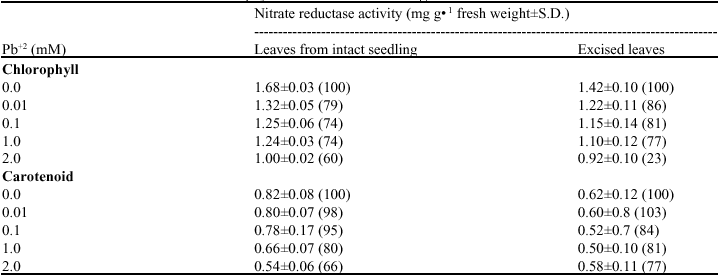
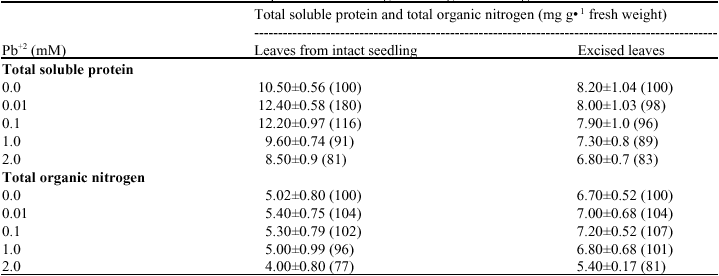
Effect of Lead on Seed Germination, Seedling Growth, Chlorophyll Content and Nitrate Reductase Activity in Mung Bean (Vigna radiata)
College of Biotechnology, Sardar Vallabh Bhai Patel University of Agriculture and Technology,Meerut-250110, India
Madhu Gautam
Institute of Management Studies, C-238, Bulandshahr Road, Indl, Area, Lal Quan, G.T. Road, Ghaziabad-201009, India
Sanjay Kumar Garg
Department of Plant Science, M.J.P. Rohilkhand University, Bareilly-243006, India
Reshu Chaudhary
College of Biotechnology, Sardar Vallabh Bhai Patel University of Agriculture and Technology,Meerut-250110, India
Kalpana Sengar
College of Biotechnology, Sardar Vallabh Bhai Patel University of Agriculture and Technology,Meerut-250110, India