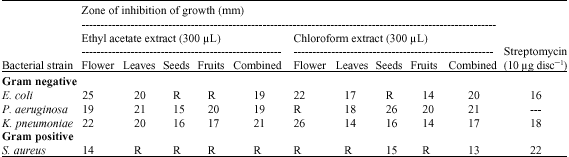
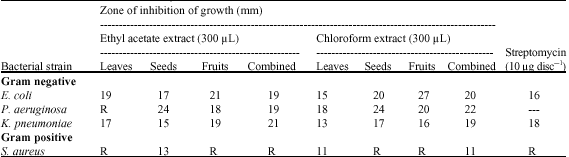
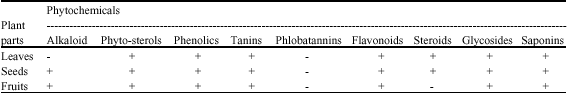
Research Article
Antibacterial Activities and Phytochemical Analysis of Different Plant Parts of Nyctanthes arbor-tristis (Linn.)
School of Biotechnology, Chemical and Biomedical Engineering, Veilore Institute of Technology University, Vellore-632 014 (T.N.), India
Deepak Ganjewala
School of Biotechnology, Chemical and Biomedical Engineering, Veilore Institute of Technology University, Vellore-632 014 (T.N.), India