Research Article
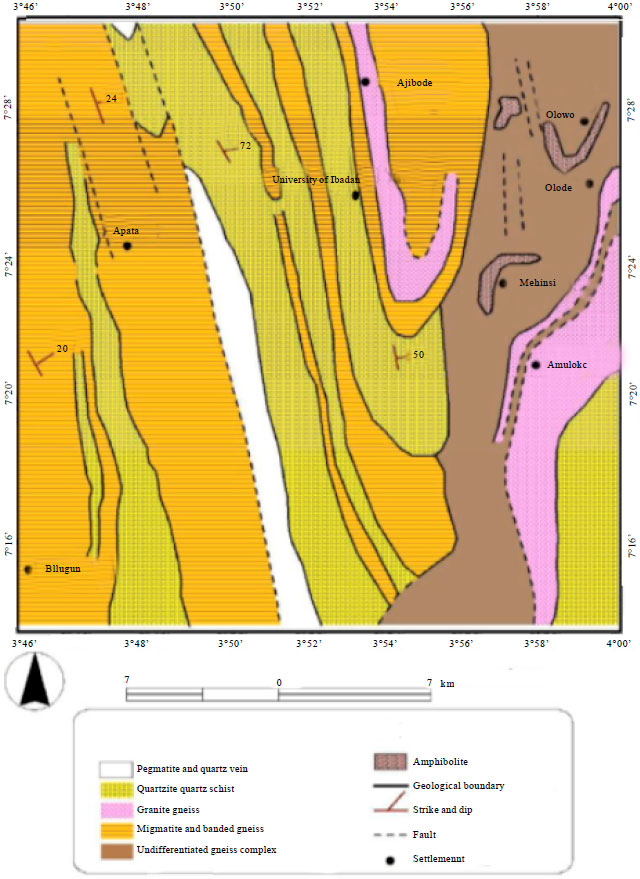
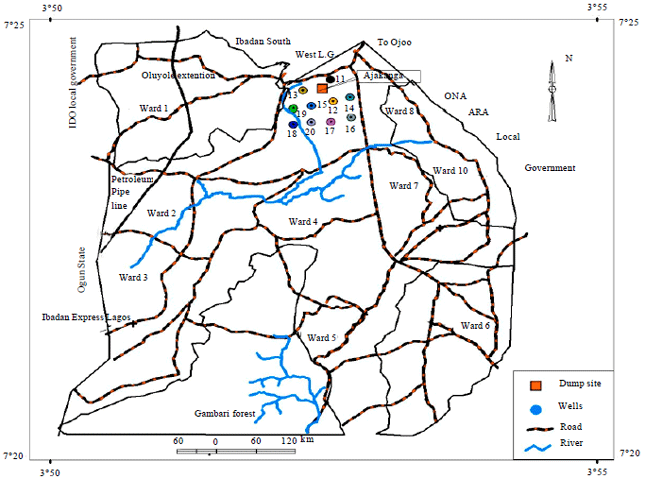
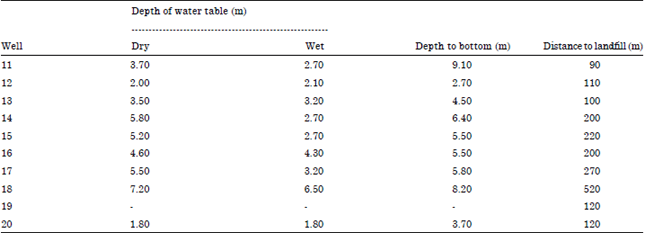
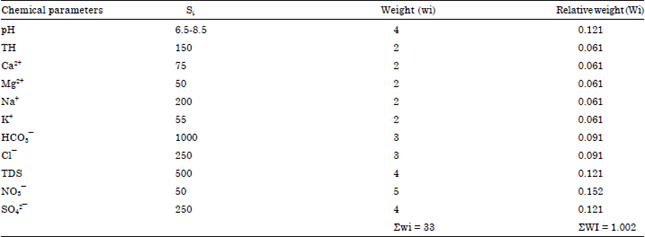
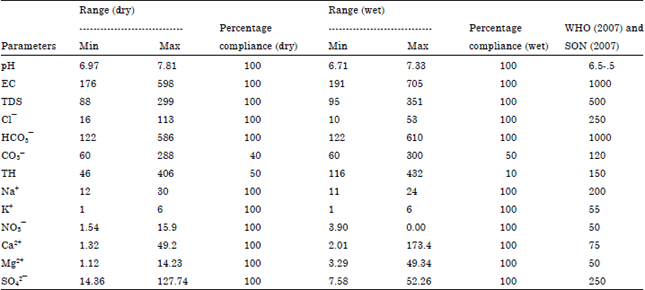
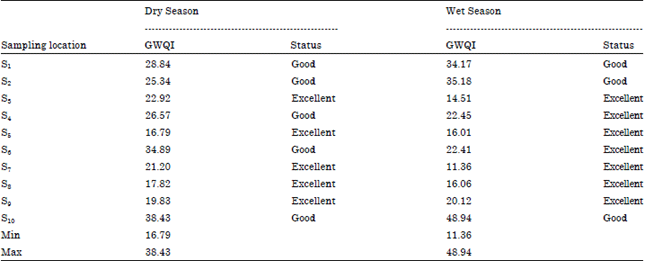
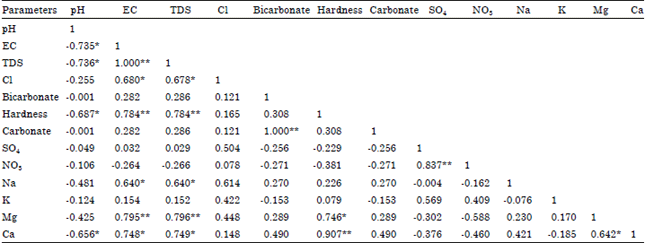
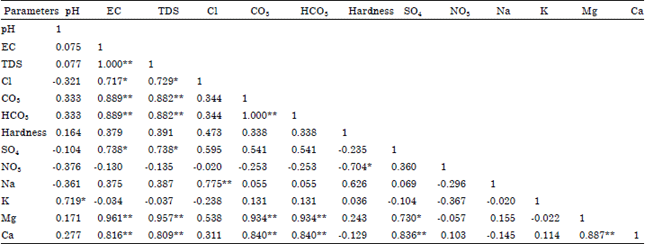
Seasonal Variations of Physico Chemical Properties and Quality Index of Groundwater of Hand-Dug Wells Around Ajakanga Dump Site in Southwestern Nigeria
Department of Physics, Federal University of Agriculture Abeokuta, P.M.B. 2240, Abeokuta, Ogun State, Nigeria
V.C. Ozebo
Department of Physics, Federal University of Agriculture Abeokuta, P.M.B. 2240, Abeokuta, Ogun State, Nigeria
O.A. Idowu
Department of Water Resources and Agrometeorology, Federal University of Agriculture, P.M.B. 2240, Abeokuta, Ogun State Nigeria
S.A. Ganiyu
Department of Physics, Federal University of Agriculture Abeokuta, P.M.B. 2240, Abeokuta, Ogun State, Nigeria
O.T. Olurin
Department of Physics, Federal University of Agriculture Abeokuta, P.M.B. 2240, Abeokuta, Ogun State, Nigeria