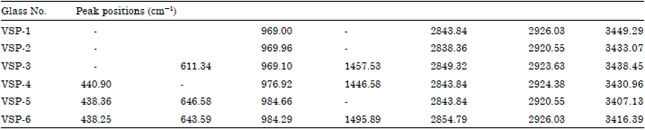
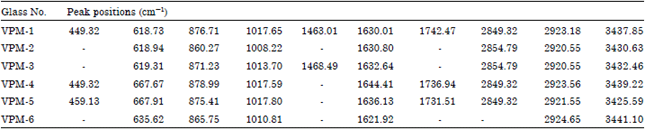
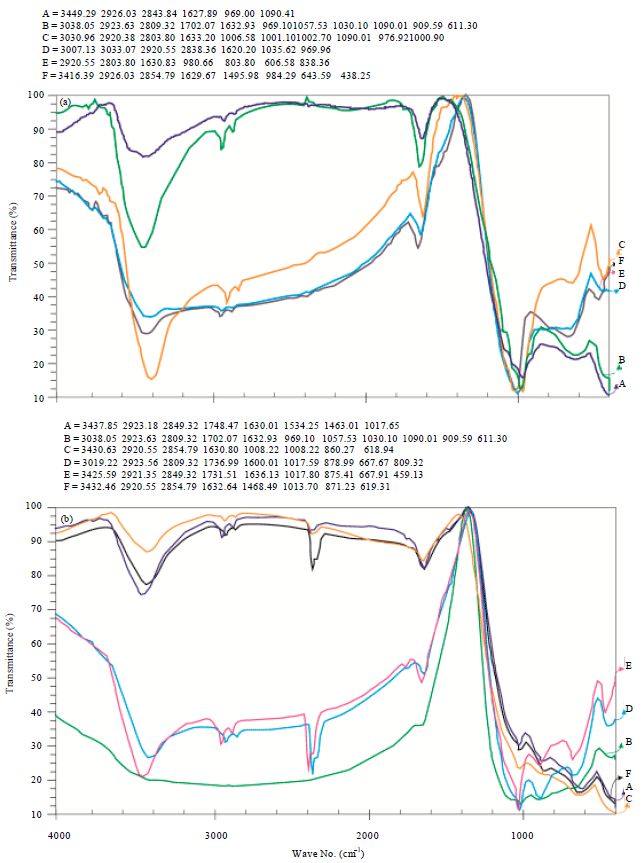
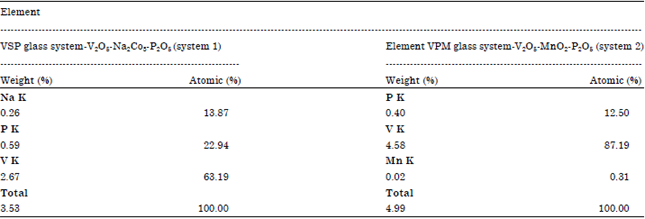
Research Article
Ultrasonic and Spectroscopic Studies on Structural Elucidation of Some Vanadium Glasses
Department of Physics [DDE], Annamalai University, Annamalai Nagar, India
J. Earnest Jayakumar
Department of Physics [DDE], Annamalai University, Annamalai Nagar, India