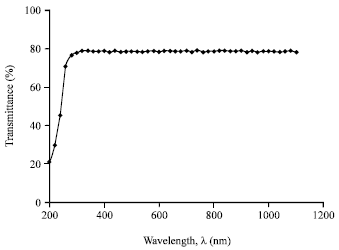
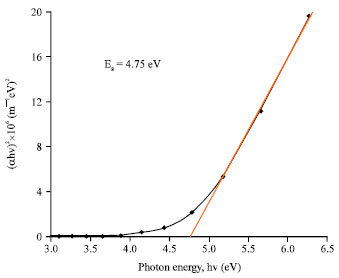
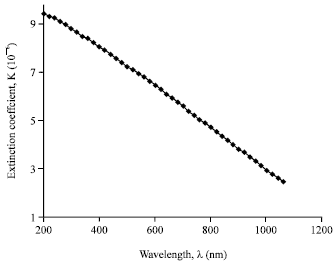
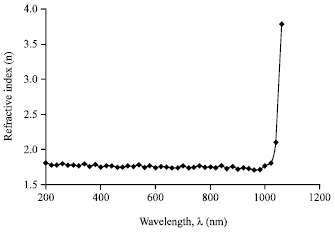
Research Article
A Study on Growth, Structural, Optical and Electrical Characterization of L-alanine Single Crystal for Optoelectronic Devices
Department of Textile Engineering, Southeast University, Banani, Bangladesh
Jiban Podder
Department of Physics, Bangladesh University of Engineering and Technology, Dhaka-1000, Bangladesh