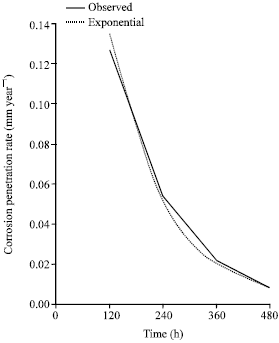
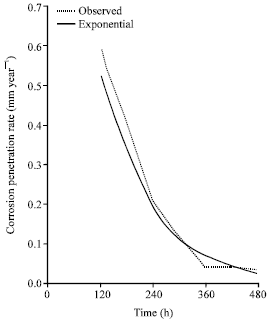
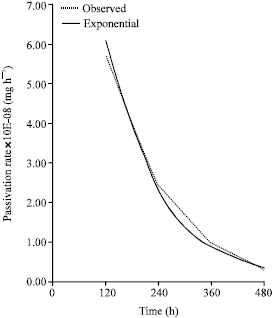
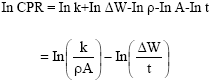Research Article
Statistical Analysis of the Influence of Environment on Prediction of Corrosion from its Parameters
Department of Industrial Physics, Faculty of Applied Natural Sciences, Ebonyi State University, P.M.B. 053 Abakaliki, Nigeria
N.E. Idenyi
Department of Industrial Physics, Faculty of Applied Natural Sciences, Ebonyi State University, P.M.B. 053 Abakaliki, Nigeria