Review Article
Effect of Cadmium on Female Reproduction and Treatment Options
Department of Biochemistry Unit, Biological Sciences, Landmark University, Omu-Aran, Kwara State, Nigeria
LiveDNA: 234.24024
Although heavy metals are naturally found in the earth crust, certain anthropogenic events have changed their normal biochemical balance, thereby increasing their accumulation in the environment. Some heavy metals are an important constituent of enzymes and are therefore required by living organisms in minute amounts; iron, molybdenum, cobalt, copper, zinc and manganese are specific examples of such metals1. Other heavy metals have no known benefit on living organisms and are known to be toxic even in small amounts, examples of these are cadmium, lead, mercury and plutonium. All heavy metals are however toxic in high concentrations due to their ability to alter enzyme function, substitute for other essential metals or generate reactive oxygen species2,3.
Cadmium is a toxic heavy metal that has no known advantageous role to the human biological system, it however poses a major public health issue. It especially has detrimental effects on the reproductive system4,5. Cadmium has been previously shown to be a developmental toxicant as well as that of the reproductive system6. It is a known endocrine disruptor due to its ability to interfere with hormonal action7. It is naturally found in the earth crust and released as a result of erosion and illegal mining activities. Cadmium is commonly found in cigarette smoke, nickel-cadmium batteries and television screens. Humans are usually exposed to cadmium through inhalation and ingestion of contaminated products8. It has a long biological half-life of 15-20 years and accumulates in the blood, kidneys, liver and the gonads with time9,10.
Generally, the reproductive system is responsible for two major events in living organisms, these are; gametogenesis and secretion of sex hormones. In females however, the reproductive system has additional functions of housing a growing fetus and delivery to the external environment. The female reproductive system is made up of the uterus, ovaries, fallopian tubes and the vagina. It is highly susceptible to damage by cadmium; the body burden of cadmium in females especially those with iron deficiency is generally higher than in males because the duodenal iron transporter is upregulated by iron deficiency which leads to an elevated absorption of dietary cadmium by the intestines11. The female reproductive system, especially the uterus can have altered or enhanced functioning due to ingestion of plant products and other chemical compounds, including cadmium. This may occur through their direct action on the uterus or through their effect on the hypothalamic-pituitary-gonadal axis, inhibiting or improving certain important biochemical processes12.
Cadmium can enter the human system by ingestion or inhalation, which could be from cigarette smoke, pollution from industries making use of cadmium as raw material, contaminated drinking water and food sources, illegal mining activities and pollution. Once cadmium enters the human system, it has a low excretion rate with biological half-life of about 20-40 years5. Its major negative effects are usually observed in the reproductive system, the kidneys and the bone. Cadmium displays the ability to accumulate in human endometrium, the lining of the uterus13.
At the cellular level, cadmium attacks the mitochondria and prevents both cellular respiration and oxidative phosphorylation even in minute quantity. Cadmium also enhances generation of reactive oxygen species and triggers apoptosis. In addition, antioxidant enzymes’ activities are depleted as a consequence of cadmium action on the cells. The overall effect of cadmium on the reproductive system of females include inhibition of ovarian function, hindering the rate of steroidogenesis, enhancement in ovarian haemorrhage and necrosis, increase in the rate of spontaneous abortions and reduction in live births rate14.
These negative effects observed as a result of cadmium intake can be remedied by the use of orthodox medicine involving chemical metal chelators like calcium sodium salt of EDTA ethylene diamine tetraacetic acid, DMSA Dimercaptosuccinic acid, DMPS dimercapto propanesulfonic acid and their likes. Recently plants and their products are being investigated for their ability to ameliorate the effect of cadmium on female reproductive system mostly because of their antioxidant properties. Phytotherapy seems to be the preferred option since the orthodox metal chelators have their side effects which range from mild to severe.
Cadmium toxicity has been a major public health issue amongst women of reproductive age and its detrimental effects on the reproductive system cannot be overemphasised. This review was therefore aimed at highlighting the effects of cadmium on the reproductive system of females and the possible remedy options available.
OVERVIEW OF CADMIUM TOXICITY
Cadmium, a soft, ductile, silvery white and bluish heavy metal that has atomic number 48, mass number 112, melting and boiling points 321 and 765°C, respectively. It has 8 isotopes which are stable, these are; 106Cd, 108Cd, 110Cd, 111Cd, 112Cd, 113Cd, 114Cd and 116Cd. Amongst these, the most common15,14 are 112Cd and 114Cd. Cadmium is released into the environment from both natural and anthropogenic activities, it acts as an environmental and occupational pollutant, posing a risk to both human and animal health.
It is found in little amount or quantity in human diets16 and mostly used in industrial procedures e.g. mining, plastic stabilizers, pigment production, nickel-cadmium battery production, refining of copper, zinc and lead ores. It is also used as a color pigment, stabilizer in PVC products, an anti-corrosive agent, in the manufacturing of nickel-cadmium batteries, metal coatings and in phosphate fertilizers fairly contains large amount of cadmium. Humans can be exposed to cadmium by direct exposure and inhalation e.g., cigarette smoking or ingestion of contaminated food or water. Cadmium is commonly found in cigarette smoke, tap water, coffee, sea foods especially shellfish, marijuana and also in some refined foods like hydrogenated oils17.
The main sources of exposure to cadmium include natural and human sources. Humans and other living organisms are naturally exposed to cadmium through air, soils and water; cadmium is released in ambient air from occupational environments especially in industries that manufacture pharmaceuticals, paints, semiconductors, audio speakers, shoes and laboratories. It is also released from tobacco smoking because tobacco leaves are naturally rich in cadmium. Cadmium is found in soils from manure, phosphate fertilizers, sewage sludge and from underlying bedrock. It is also a minor component of surface and underground water where erosions can lead to its release to living organisms. Human sources of cadmium exposure include illegal mining activities, electroplating, smelting and refining of zinc, copper and lead ores, production of nickel-cadmium batteries and welding18,19.
For all non-smoking adults in the US, the national geometric mean blood cadmium levels is 0.47 μg L–1 while it is approximately double at 1.58 μg L–1 in smokers20. The agency for toxic substances and disease registry (ATSDR) has however fixed the geometric blood level of cadmium in the general population at 0.315 μg L–1 while the geometric urine level of cadmium in the general population21 is 0.185 μg L–1. The maximum contaminant level MCL of cadmium in drinking water22 is 0.005 μg L–1. Although cadmium is a significant public health issue in Nigeria, yet poor records keeping as well as absence of poison information centers are major characteristics of Nigerian environmental health23.
Cadmium concentrations in the human system can be detected through blood analysis, urine analysis and hair analysis. The blood analysis helps to detect recent exposure to cadmium while urine analysis detects accurately both recent and past exposure to cadmium24.
After ingestion of cadmium, it is taken up by the blood where it binds to proteins such as metallothionein and albumin. It is then first transported to the liver after its uptake into the gastrointestinal tract. In the liver, cadmium triggers hepatocyte necrosis and apoptosis, cadmium also induces the production of metallothionein in the liver, complexes of cadmium-metallothionein formed are subsequently washed into the blood. Subsequently, some of the cadmium already absorbed enters the enterohepatic cycle through discharge into the biliary tracts as cadmium-glutathione conjugates, this is broken down to cadmium-cysteine complexes. Cadmium can then re-enter the small intestines25. The second main target organ for cadmium is the kidney where it is accumulated for a long term leading to tubulus cell necrosis. Cadmium can then be excreted through urine and faeces26,16.
Cadmium toxicity affects multiple organ systems, this comprise the lungs, the kidneys, liver, skeletal system, reproductive system and the cardiovascular systems27. Cadmium is grouped as a class I human carcinogen by the International Agency for Research on Cancer (IARC)9. There are several proposed mechanisms for the toxic action of cadmium which include the ability to displace vital minerals from their native enzymes and other biomolecules thereby hindering the biological activity of such compounds, oxidative stress is another major mechanism of toxicity2.
Cadmium and female reproductive system: In the female reproductive system, cadmium is known to have the following effects: alteration of steroidogenesis, delayed puberty and menarche, pregnancy loss, disorders of the menstrual cycle and reproductive hormones, premature birth and reduced birth weights.
Cholesterol is known as the main precursor of the female sex hormones estrogen and progesterone. Cadmium has demonstrated capacity in disrupting the rates of steroidogenesis in the ovaries and placenta, thereby preventing the secretion of the female sex hormones and consequently adversely affecting normal reproduction28. Cadmium impedes progesterone synthesis by inhibiting the expression of steroidogenic acute regulatory protein StAR and cytochrome P450 cholesterol side chain enzyme P450scc. The StAR protein mediates intra mitochondrial transport of cholesterol29, while the P450scc is an enzyme of the mitochondrial membrane that has the responsibility of converting cholesterol to pregnenolone, this is the first step in the steroidogenic pathway in mammalian tissues. Cadmium also builds up in the granulosa cells of the ovary leading to a significant decrease in the gonadotropins thereby disrupting the activities of steroidogenic enzymes30,31.
Cadmium can suppress the pregnancy hormone, progesterone preventing its role of sustaining pregnancy especially in the first 13 weeks of gestation leading to miscarriages32. Miscarriage is also called spontaneous abortion; it is defined as a pregnancy that typically terminates naturally without induction during the first 7-28 weeks of gestation. It is a known public health challenge in developing countries33. Cadmium can cause miscarriages through its ability to cross the placental barrier, accumulate in fetal tissues thereby hindering the major functions of the placental; like hampering its roles of signaling, transport of nutrients, cellular growth and maturation, secretion of hormones and enzymes34-36. Maternal exposure to high levels of cadmium has also been linked with decreased birth weights and premature births. Cadmium absorbed by the body is deposited in the placenta, this may enter into fetal circulation thereby blocking nutrients and blood flow resulting into growth retardation37.
High level of cadmium in humans has been associated with delay in puberty and/or menarche, this may be because cadmium inhibits estrogen activity, cadmium also reduces the secretion of inhibin B, which is a hormone secreted by the granulosa cells of the ovaries that acts majorly to suppress the synthesis and secretion of the follicle stimulating hormone by the anterior pituitary gland32. Studies carried out on the effect of in-utero cadmium exposure have reported that high doses of exposure to cadmium, may delay androgenic development, the first stage of puberty and menarche, this may probably be due to delayed onset of estrus as a result of ovotoxicity38.
Normally, the menstrual cycle is regulated by both the nervous and endocrine hormone system in a regular monthly series of events. Cadmium can damage the nerve cells and/or inhibit the synthesis and secretion of the female reproductive hormones resulting in menstrual disorders39. Exposure to cadmium has been linked to alterations in the activity of the endocrine system in females. Even at very low levels, cadmium affects circadian variation and concentration of pituitary hormones in rats’ plasma. It also affects circulating levels of growth, prolactin and adrenocorticotropic hormones40.
Mechanisms of action for cadmium toxicity: Although cadmium on its own cannot generate reactive oxygen species directly, it generates non-radical hydrogen peroxide, which is subsequently a source of free radicals generation through the Fenton reaction41,42. Cadmium induces oxidative stress indirectly by decreasing intracellular glutathione levels, combining with thiol groups of antioxidant enzymes, e.g., catalase, glutathione peroxidase and superoxide dismutase thereby hindering their normal functions.
Cadmium also obstructs complex III of the mitochondrial electron transport chain thereby enhancing the production of reactive oxygen species ROS resulting into negative alteration of mitochondrial membrane and subsequently encouraging apoptosis43. Cadmium has the ability to replace iron, zinc, copper, magnesium and calcium from a number of biomolecules and membrane proteins. This can alter the function of such biomolecules and cause an increase in the levels of such metals, this can be linked in some cases to production of oxidative stress through the Fenton reaction. Cadmium has the ability to bind up to ten times more strongly than zinc and is much more difficult to remove43.
Cadmium has high affinity for selenium and easily binds with it, the complex formed is subsequently excreted through the bile system. This results into the unavailability of selenium to form glutathione peroxidase, one of the system’s primary antioxidants43. Cadmium interferes with the DNA binding zinc finger motif by substituting cadmium ion for zinc ion. It also acts as an endocrine disruptor mimicking and hindering the effects of endogenous oestrogens thereby impeding steroidogenesis9.
Figure 1 shows the suppression of the cellular antioxidant system by cadmium, indirectly activating oxidative stress. Cadmium prevents expression of the major antioxidants like glutathione (GSH), Superoxide dismutase (SOD) and catalase (CAT) leading to increased levels of reactive oxygen species (ROS) generation and lipid peroxidation44.
Cadmium poisoning remediation in humans: Although there are no specific therapies directed at treating the reproductive effect of cadmium poisoning in humans. However, generally cadmium poisoning has been remedied with orthodox medicines, involving the use of metal chelators which can be taken orally or administered intravenously.
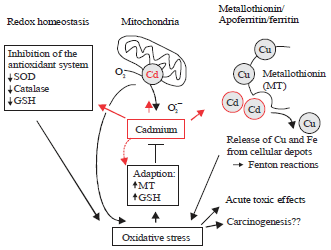 | |
| Fig. 1: | Cadmium suppression of the cellular antioxidant system (Adapted from Henkler et al.44) |
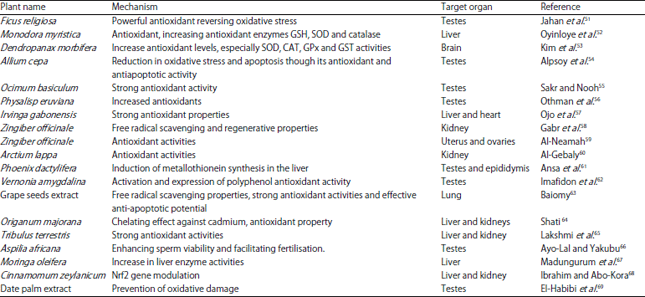
| Table 1: | Mechanisms of action and target organs of plants with protective effect against cadmium-induced toxicity |
 | |
In recent years, antioxidants supplementations and the use of plants are being explored and have been therapeutic against the detrimental effects of cadmium on living organisms.
The treatment options for cadmium poisoning include the use of orthodox medicines which act as chelators, binding heavy metals in the blood and delivering these to the excretory system for safe disposal of the toxic substance. This comprises the use of calcium disodium ethylene diamine tetraacetic acid EDTA, 2,3-dimercaptopropanesulfonic acid DMPS and Dimercaptosuccinic acid DMSA. Intravenous chelation with EDTA is however regarded as the most widely accepted method of cadmium detoxification for clinical use when carried out following established protocols45.
Vitamins and minerals supplementation has also been used previously to prevent and curb the effect of cadmium poisoning on living organisms46-48.
Cadmium has similar physical and chemical characteristics with zinc. Therefore, zinc administered to patients with high level of blood cadmium can displace cadmium from the usual binding sites of zinc in metalloproteins and enzymatic antioxidants thereby eliminating cadmium from the tissues. Induction of metallothionein synthesis is also enhanced through the intake of zinc. Metallothionein is a low molecular weight protein localized in the membrane of the golgi apparatus, it has high affinity for cadmium and other heavy metals through the thiol group of its cysteine residues thereby helping to detoxify them. Zinc can prevent oxidative stress due to its ability to function as a co-factor of the antioxidant enzyme copper zinc superoxide dismutase27 Cu/Zn SOD.
Some plants have been studied for their protective effect on cadmium-induced toxicity in rats. The treatment of cadmium-exposed female Wistar rats with a preparation of 17 different herbs was reported to reverse the decrease in level of anti-oxidant enzymes. This thereby enhanced the levels of antioxidant enzymes in the ovaries of the female Wistar rats49. Aqueous seed extracts of Foeniculum vulgare and Carumcarvi have been shown to ameliorate the effect of cadmium-induced gonadal damage in female albino rats50. Other plants that have previously demonstrated protective effects against cadmium-induced toxicity are displayed in Table 1.
This study highlighted the effects of cadmium on the reproductive system of females, the mechanisms of action and the remedy options available. The detrimental effects of cadmium toxicity include alteration of steroidogenesis, delayed puberty and menarche, pregnancy loss, disorders of the menstrual cycle and reproductive hormones, premature birth and reduced birth weights. The mechanisms by which cadmium exerts these effects are through the ability to displace vital minerals from their native enzymes, thereby hindering the biological activity of such enzymes, oxidative stress is another major mechanism of cadmium toxicity.
Although there are no specific therapies directed at treating only the reproductive effect of cadmium poisoning in humans, however, this current review highlighted the treatment options for cadmium-induced toxicity. This includes the use of orthodox medicines, vitamins and minerals supplementation as well as phytoremediation.